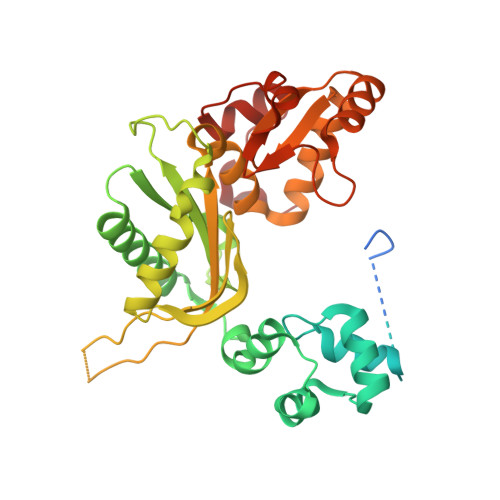
Structures of the Leishmania infantum polymerase beta.
Mejia, E., Burak, M., Alonso, A., Larraga, V., Kunkel, T.A., Bebenek, K., Garcia-Diaz, M.(2014) DNA Repair (Amst) 18: 1-9
- PubMed: 24666693 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.dnarep.2014.03.001
- Primary Citation Related Structures:
4P4M, 4P4O, 4P4P - PubMed Abstract:
Protozoans of the genus Leishmania, the pathogenic agent causing leishmaniasis, encode the family X DNA polymerase Li Pol β. Here, we report the first crystal structures of Li Pol β. Our pre- and post-catalytic structures show that the polymerase adopts the common family X DNA polymerase fold. However, in contrast to other family X DNA polymerases, the dNTP-induced conformational changes in Li Pol β are much more subtle. Moreover, pre- and post-catalytic structures reveal that Li Pol β interacts with the template strand through a nonconserved, variable region known as loop3. Li Pol β Δloop3 mutants display a higher catalytic rate, catalytic efficiency and overall error rates with respect to WT Li Pol β. These results further demonstrate the subtle structural variability that exists within this family of enzymes and provides insight into how this variability underlies the substantial functional differences among their members.
- Department of Pharmacological Sciences, Stony Brook University, BST 7-169, Stony Brook, NY 11794-8651, USA.
Organizational Affiliation: