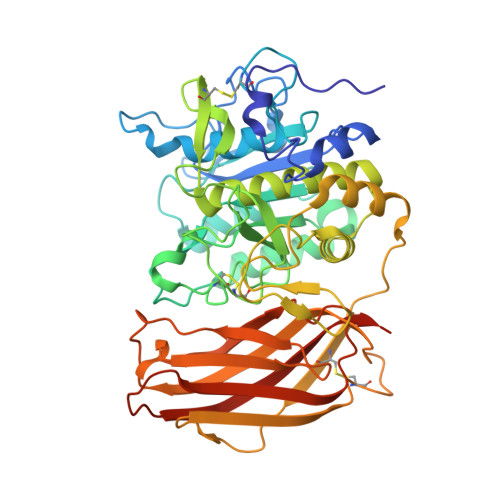
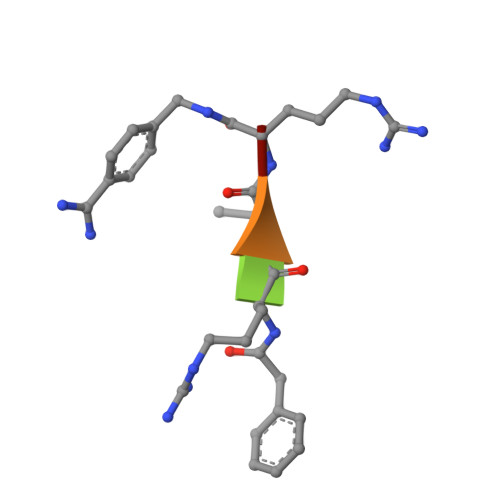
X-ray Structures of Human Furin in Complex with Competitive Inhibitors.
Dahms, S.O., Hardes, K., Becker, G.L., Steinmetzer, T., Brandstetter, H., Than, M.E.(2014) ACS Chem Biol 9: 1113-1118
- PubMed: 24666235
- DOI: https://doi.org/10.1021/cb500087x
- Primary Citation of Related Structures:
4OMC, 4OMD - PubMed Abstract:
Furin inhibitors are promising therapeutics for the treatment of cancer and numerous infections caused by bacteria and viruses, including the highly lethal Bacillus anthracis or the pandemic influenza virus. Development and improvement of inhibitors for pharmacological use require a detailed knowledge of the protease's substrate and inhibitor binding properties. Here we present a novel preparation of human furin and the first crystal structures of this enzyme in complex with noncovalent inhibitors. We show the inhibitor exchange by soaking, allowing the investigation of additional inhibitors and substrate analogues. Thus, our work provides a basis for the rational design of furin inhibitors.
- Protein Crystallography Group, Leibniz Institute for Age Research-Fritz Lipmann Institute (FLI) , Beutenbergstr. 11, 07745 Jena, Germany.
Organizational Affiliation: