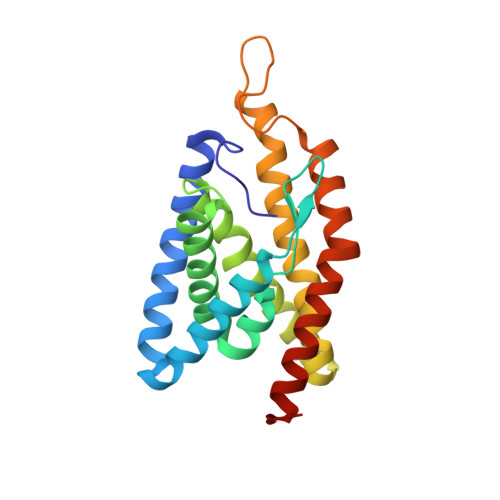
Planar substrate-binding site dictates the specificity of ECF-type nickel/cobalt transporters
Yu, Y., Zhou, M.Z., Kirsch, F., Xu, C.Q., Zhang, L., Wang, Y., Jiang, Z., Wang, N., Li, J., Eitinger, T., Yang, M.J.(2014) Cell Res 24: 267-277
- PubMed: 24366337 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/cr.2013.172
- Primary Citation Related Structures:
4M58, 4M5B, 4M5C - PubMed Abstract:
The energy-coupling factor (ECF) transporters are multi-subunit protein complexes that mediate uptake of transition-metal ions and vitamins in about 50% of the prokaryotes, including bacteria and archaea. Biological and structural studies have been focused on ECF transporters for vitamins, but the molecular mechanism by which ECF systems transport metal ions from the environment remains unknown. Here we report the first crystal structure of a NikM, TtNikM2, the substrate-binding component (S component) of an ECF-type nickel transporter from Thermoanaerobacter tengcongensis. In contrast to the structures of the vitamin-specific S proteins with six transmembrane segments (TSs), TtNikM2 possesses an additional TS at its N-terminal region, resulting in an extracellular N-terminus. The highly conserved N-terminal loop inserts into the center of TtNikM2 and occludes a region corresponding to the substrate-binding sites of the vitamin-specific S components. Nickel binds to NikM via its coordination to four nitrogen atoms, which are derived from Met1, His2 and His67 residues. These nitrogen atoms form an approximately square-planar geometry, similar to that of the metal ion-binding sites in the amino-terminal Cu(2+)- and Ni(2+)-binding (ATCUN) motif. Replacements of residues in NikM contributing to nickel coordination compromised the Ni-transport activity. Furthermore, systematic quantum chemical investigation indicated that this geometry enables NikM to also selectively recognize Co(2+). Indeed, the structure of TtNikM2 containing a bound Co(2+) ion has almost no conformational change compared to the structure that contains a nickel ion. Together, our data reveal an evolutionarily conserved mechanism underlying the metal selectivity of EcfS proteins, and provide insights into the ion-translocation process mediated by ECF transporters.
- 1] MOE Key Laboratory of Protein Sciences, Tsinghua-Peking Center for Life Sciences, School of Life Sciences, Tsinghua University, Beijing 100084, China [2] Department of Pharmacology and Pharmaceutical Sciences, School of Medicine, Tsinghua University, Beijing 100084, China.
Organizational Affiliation: