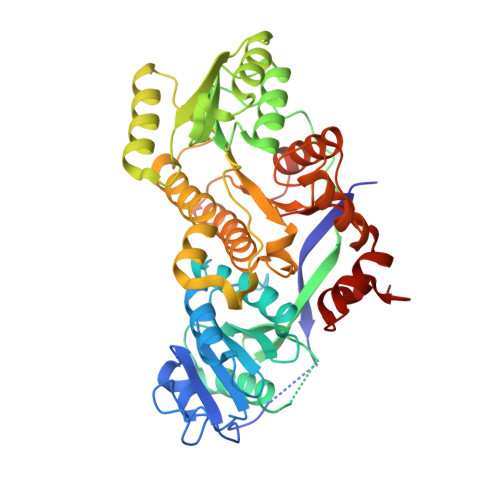
Structural and Stereochemical Analysis of a Modular Polyketide Synthase Ketoreductase Domain Required for the Generation of a cis-Alkene.
Bonnett, S.A., Whicher, J.R., Papireddy, K., Florova, G., Smith, J.L., Reynolds, K.A.(2013) Chem Biol 20: 772-783
- PubMed: 23790488 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.chembiol.2013.04.014
- Primary Citation Related Structures:
4HXY - PubMed Abstract:
The formation of an activated cis-3-cyclohexylpropenoic acid by Plm1, the first extension module of the phoslactomycin polyketide synthase, is proposed to occur through an L-3-hydroxyacyl-intermediate as a result of ketoreduction by an A-type ketoreductase (KR). Here, we demonstrate that the KR domain of Plm1 (PlmKR1) catalyzes the formation of an L-3-hydroxyacyl product. The crystal structure of PlmKR1 revealed a well-ordered active site with a nearby Trp residue characteristic of A-type KRs. Structural comparison of PlmKR1 with B-type KRs that produce D-3-hydroxyacyl intermediates revealed significant differences. The active site of cofactor-bound A-type KRs is in a catalysis-ready state, whereas cofactor-bound B-type KRs are in a precatalytic state. Furthermore, the closed lid loop in substrate-bound A-type KRs restricts active site access from all but one direction, which is proposed to control the stereochemistry of ketoreduction.
- Department of Chemistry, Portland State University, Portland, OR. 97201.
Organizational Affiliation: