Crystal structure of human WD repeat domain 5 with compound MM-401
Karatas, H., Townsend, E.C., Chen, Y., Bernard, D., Liu, L., Dou, Y., Lei, M., Wang, S.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| WD repeat-containing protein 5 | 313 | Homo sapiens | Mutation(s): 0 Gene Names: WDR5, BIG3 |  | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P61964 GTEx: ENSG00000196363 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P61964 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| MM-401 | C [auth E], D [auth F] | 5 | N/A | Mutation(s): 0 | 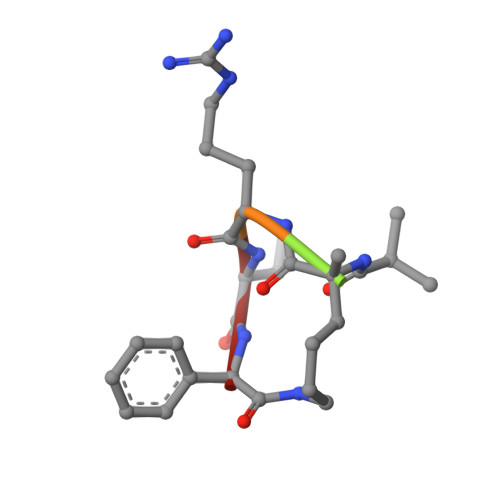 |
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| 0XO Query on 0XO | C [auth E], D [auth F] | D-PEPTIDE LINKING | C7 H16 N2 O2 |  | -- |
| ABA Query on ABA | C [auth E], D [auth F] | L-PEPTIDE LINKING | C4 H9 N O2 |  | ALA |
| PG9 Query on PG9 | C [auth E], D [auth F] | D-PEPTIDE LINKING | C8 H9 N O2 |  | -- |
| Entity ID: 2 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_000897 Query on PRD_000897 | C [auth E], D [auth F] | MM-401 | Peptide-like / Inhibitor |  | |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 46.904 | α = 88.5 |
| b = 47.242 | β = 89.5 |
| c = 68.896 | γ = 74.53 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data collection |
| PHASER | phasing |
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |