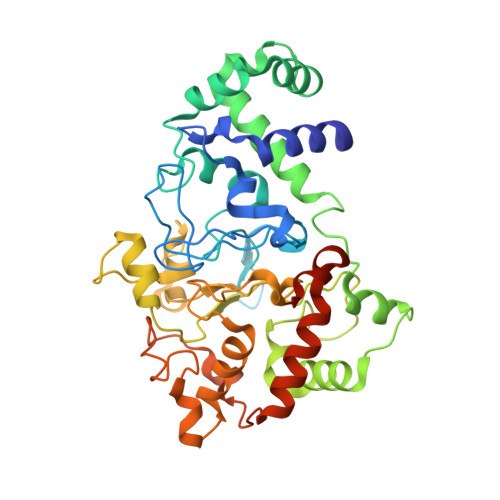
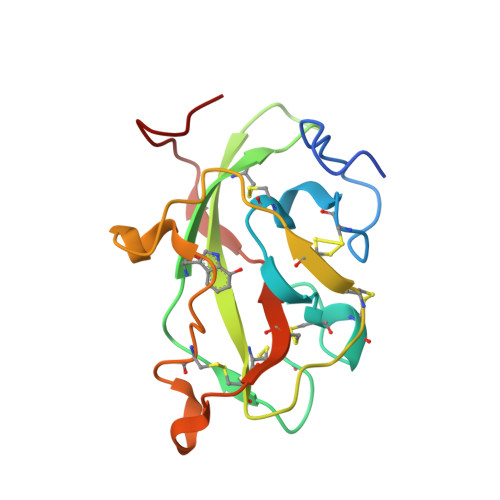
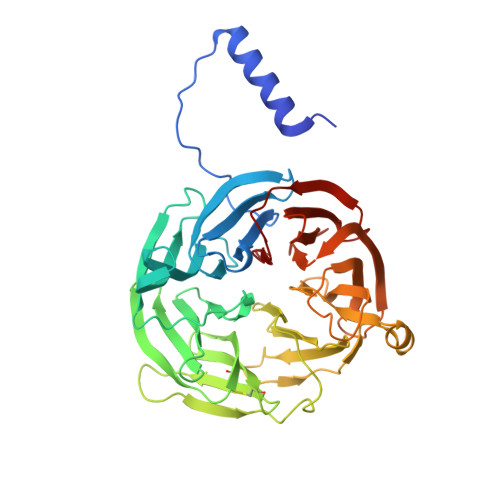
Diradical intermediate within the context of tryptophan tryptophylquinone biosynthesis.
Yukl, E.T., Liu, F., Krzystek, J., Shin, S., Jensen, L.M., Davidson, V.L., Wilmot, C.M., Liu, A.(2013) Proc Natl Acad Sci U S A 110: 4569-4573
- PubMed: 23487750 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1215011110
- Primary Citation Related Structures:
4FA1, 4FA4, 4FA5, 4FA9, 4FAN, 4FAV, 4FB1 - PubMed Abstract:
Despite the importance of tryptophan (Trp) radicals in biology, very few radicals have been trapped and characterized in a physiologically meaningful context. Here we demonstrate that the diheme enzyme MauG uses Trp radical chemistry to catalyze formation of a Trp-derived tryptophan tryptophylquinone cofactor on its substrate protein, premethylamine dehydrogenase. The unusual six-electron oxidation that results in tryptophan tryptophylquinone formation occurs in three discrete two-electron catalytic steps. Here the exact order of these oxidation steps in the processive six-electron biosynthetic reaction is determined, and reaction intermediates are structurally characterized. The intermediates observed in crystal structures are also verified in solution using mass spectrometry. Furthermore, an unprecedented Trp-derived diradical species on premethylamine dehydrogenase, which is an intermediate in the first two-electron step, is characterized using high-frequency and -field electron paramagnetic resonance spectroscopy and UV-visible absorbance spectroscopy. This work defines a unique mechanism for radical-mediated catalysis of a protein substrate, and has broad implications in the areas of applied biocatalysis and understanding of oxidative protein modification during oxidative stress.
- Department of Biochemistry, Molecular Biology and Biophysics, University of Minnesota, Minneapolis, MN 55455, USA.
Organizational Affiliation: