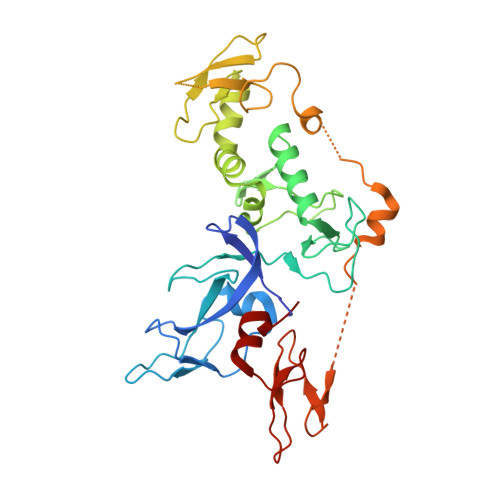
Structure of the Human Parkin Ligase Domain in an Autoinhibited State.
Wauer, T., Komander, D.(2013) EMBO J 32: 2099
- PubMed: 23727886 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/emboj.2013.125
- Primary Citation Related Structures:
4BM9 - PubMed Abstract:
Mutations in the protein Parkin are associated with Parkinson's disease (PD), the second most common neurodegenerative disease in men. Parkin is an E3 ubiquitin (Ub) ligase of the structurally uncharacterized RING-in-between-RING(IBR)-RING (RBR) family, which, in an HECT-like fashion, forms a catalytic thioester intermediate with Ub. We here report the crystal structure of human Parkin spanning the Unique Parkin domain (UPD, also annotated as RING0) and RBR domains, revealing a tightly packed structure with unanticipated domain interfaces. The UPD adopts a novel elongated Zn-binding fold, while RING2 resembles an IBR domain. Two key interactions keep Parkin in an autoinhibited conformation. A linker that connects the IBR with the RING2 over a 50-Å distance blocks the conserved E2∼Ub binding site of RING1. RING2 forms a hydrophobic interface with the UPD, burying the catalytic Cys431, which is part of a conserved catalytic triad. Opening of intra-domain interfaces activates Parkin, and enables Ub-based suicide probes to modify Cys431. The structure further reveals a putative phospho-peptide docking site in the UPD, and explains many PD-causing mutations.
- Division of Protein and Nucleic Acid Chemistry, Medical Research Council Laboratory of Molecular Biology, Cambridge, UK.
Organizational Affiliation: