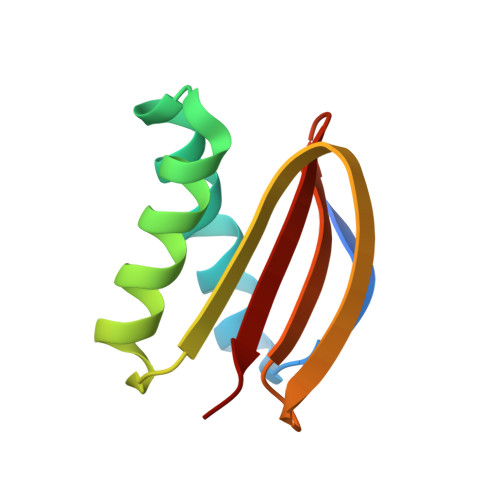
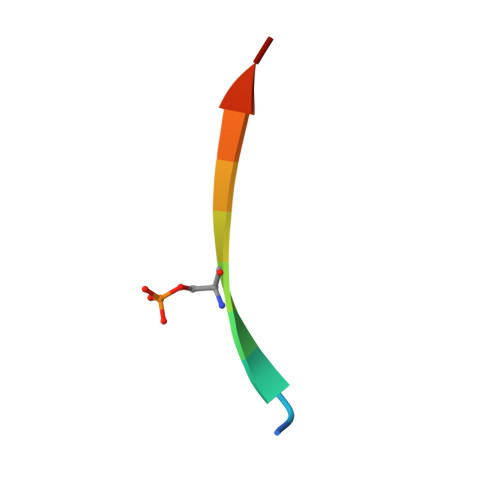
Structural Analysis of the Regulation of the Dynll/Lc8 Binding to Nek9 by Phosphorylation
Gallego, P., Velazquez-Campoy, A., Regue, L., Roig, J., Reverter, D.(2013) J Biological Chem 288: 12283
- PubMed: 23482567
- DOI: https://doi.org/10.1074/jbc.M113.459149
- Primary Citation of Related Structures:
3ZKE, 3ZKF - PubMed Abstract:
The NIMA family protein kinases Nek9/Nercc1, Nek6, and Nek7 constitute a signaling module activated in early mitosis involved in the control of spindle organization. DYNLL/LC8 (dynein light chain 8) was originally described as a component of the dynein complex, but the recent discovery of multiple interaction partners for LC8 has suggested that it has a general role as a dimerization hub that organizes different protein partners. Recent experiments suggested that LC8 binding to Nek9 was regulated by Nek9 autophosphorylation on Ser(944), a residue immediately located N-terminal to the LC8 conserved (K/R)xTQT binding motif, and that this was crucial for the control of signal transduction through the Nek/Nek6/7 module. In the present work, we present two crystal structures of LC8 with a peptide corresponding to the Nek9 binding region with and without a phosphorylation on Ser(944). Structural analysis of LC8 with both Nek9 peptides, together with different biophysical experiments, explains the observed diminished binding affinity of Nek9 to LC8 upon phosphorylation on Ser(944) within the Nek9 sequence, thus shedding light into a novel phosphorylation regulatory mechanism that interferes with LC8 protein · protein complex formation.
- Structural Biology Unit, Institut de Biotecnologia i Biomedicina, Universitat Autònoma de Barcelona, 08193 Bellaterra, Spain.
Organizational Affiliation: