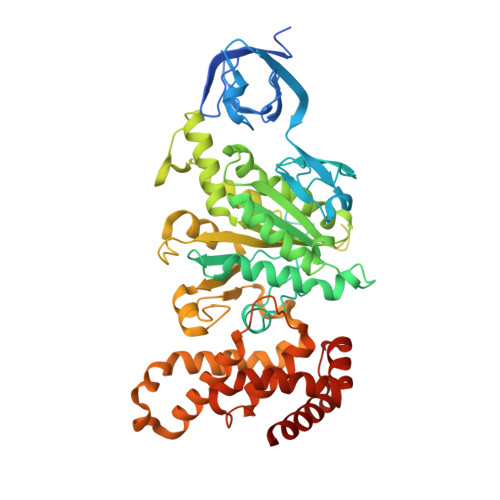
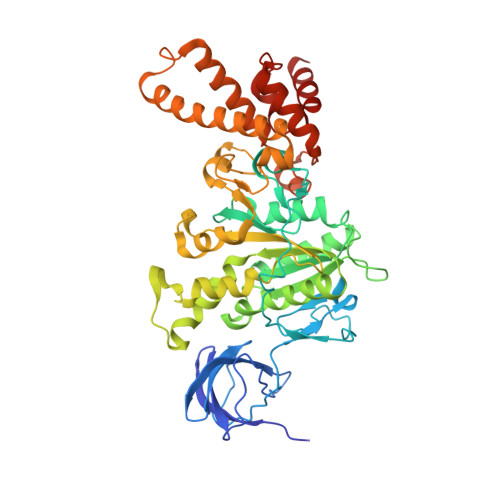
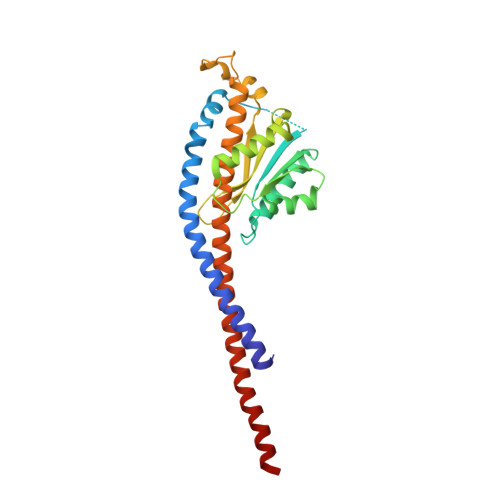
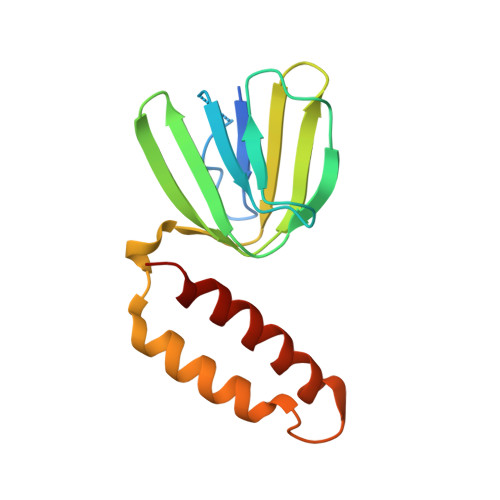
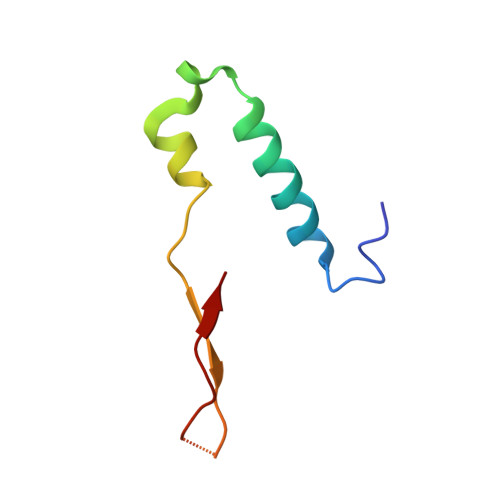
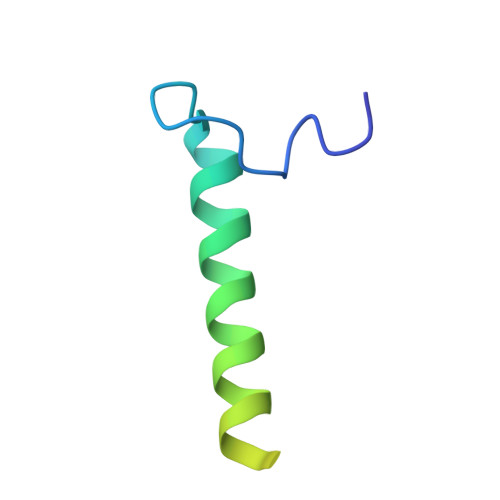
The Structure of F1-ATPase from Saccharomyces Cerevisiae Inhibited by its Regulatory Protein If1.
Robinson, G.C., Bason, J.V., Montgomery, M.G., Fearnley, I.M., Mueller, D.M., Leslie, A.G.W., Walker, J.E.(2013) Open Biol 3: 164
- PubMed: 23407639 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1098/rsob.120164
- Primary Citation Related Structures:
3ZIA - PubMed Abstract:
The structure of F₁-ATPase from Saccharomyces cerevisiae inhibited by the yeast IF₁ has been determined at 2.5 Å resolution. The inhibitory region of IF₁ from residues 1 to 36 is entrapped between the C-terminal domains of the α(DP)- and β(DP)-subunits in one of the three catalytic interfaces of the enzyme. Although the structure of the inhibited complex is similar to that of the bovine-inhibited complex, there are significant differences between the structures of the inhibitors and their detailed interactions with F₁-ATPase. However, the most significant difference is in the nucleotide occupancy of the catalytic β(E)-subunits. The nucleotide binding site in β(E)-subunit in the yeast complex contains an ADP molecule without an accompanying magnesium ion, whereas it is unoccupied in the bovine complex. Thus, the structure provides further evidence of sequential product release, with the phosphate and the magnesium ion released before the ADP molecule.
- The Medical Research Council Mitochondrial Biology Unit, Hills Road, Cambridge CB2 0XY, UK.
Organizational Affiliation: