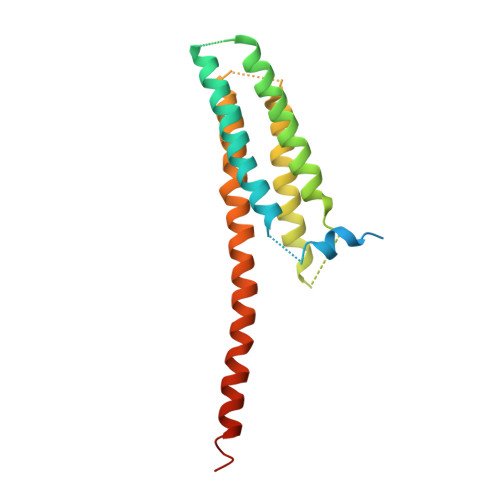
X-ray crystal structure of voltage-gated proton channel.
Takeshita, K., Sakata, S., Yamashita, E., Fujiwara, Y., Kawanabe, A., Kurokawa, T., Okochi, Y., Matsuda, M., Narita, H., Okamura, Y., Nakagawa, A.(2014) Nat Struct Mol Biol 21: 352-357
- PubMed: 24584463 Search on PubMed
- DOI: https://doi.org/10.1038/nsmb.2783
- Primary Citation Related Structures:
3WKV - PubMed Abstract:
The voltage-gated proton channel Hv1 (or VSOP) has a voltage-sensor domain (VSD) with dual roles of voltage sensing and proton permeation. Its gating is sensitive to pH and Zn(2+). Here we present a crystal structure of mouse Hv1 in the resting state at 3.45-Å resolution. The structure showed a 'closed umbrella' shape with a long helix consisting of the cytoplasmic coiled coil and the voltage-sensing helix, S4, and featured a wide inner-accessible vestibule. Two out of three arginines in S4 were located below the phenylalanine constituting the gating charge-transfer center. The extracellular region of each protomer coordinated a Zn(2+), thus suggesting that Zn(2+) stabilizes the resting state of Hv1 by competing for acidic residues that otherwise form salt bridges with voltage-sensing positive charges on S4. These findings provide a platform for understanding the general principles of voltage sensing and proton permeation.
- 1] Institute for Protein Research, Osaka University, Suita, Japan. [2] Graduate School of Medicine, Osaka University, Suita, Japan. [3] Institute for Academic Initiatives, Osaka University, Suita, Japan.
Organizational Affiliation: