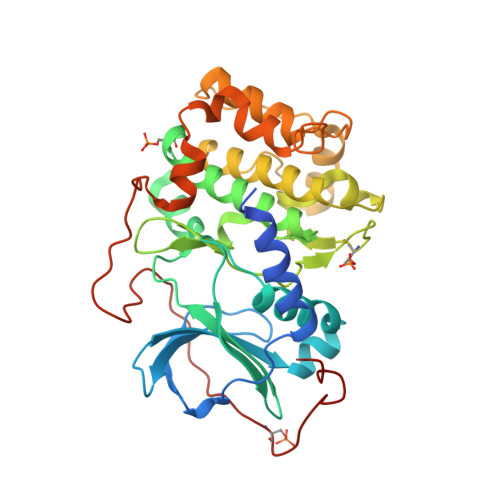
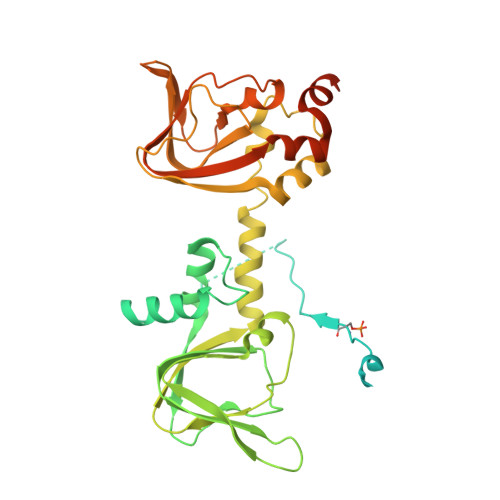
Structure and allostery of the PKA RIIbeta tetrameric holoenzyme
Zhang, P., Smith-Nguyen, E.V., Keshwani, M.M., Deal, M.S., Kornev, A.P., Taylor, S.S.(2012) Science 335: 712-716
- PubMed: 22323819 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/science.1213979
- Primary Citation Related Structures:
3TNP, 3TNQ - PubMed Abstract:
In its physiological state, cyclic adenosine monophosphate (cAMP)-dependent protein kinase (PKA) is a tetramer that contains a regulatory (R) subunit dimer and two catalytic (C) subunits. We describe here the 2.3 angstrom structure of full-length tetrameric RIIβ(2):C(2) holoenzyme. This structure showing a dimer of dimers provides a mechanistic understanding of allosteric activation by cAMP. The heterodimers are anchored together by an interface created by the β4-β5 loop in the RIIβ subunit, which docks onto the carboxyl-terminal tail of the adjacent C subunit, thereby forcing the C subunit into a fully closed conformation in the absence of nucleotide. Diffusion of magnesium adenosine triphosphate (ATP) into these crystals trapped not ATP, but the reaction products, adenosine diphosphate and the phosphorylated RIIβ subunit. This complex has implications for the dissociation-reassociation cycling of PKA. The quaternary structure of the RIIβ tetramer differs appreciably from our model of the RIα tetramer, confirming the small-angle x-ray scattering prediction that the structures of each PKA tetramer are different.
- Howard Hughes Medical Institute, University of California, San Diego, La Jolla, CA 92093-0654, USA.
Organizational Affiliation: