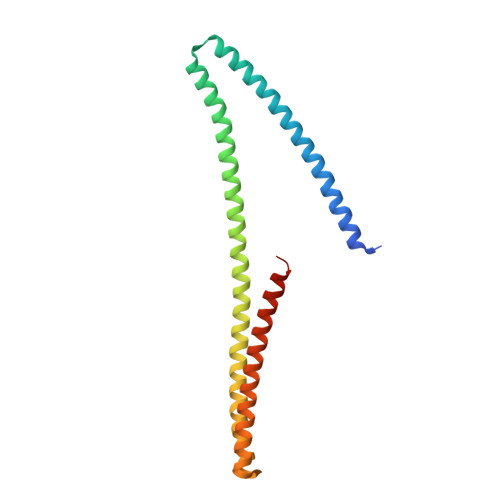Structure of Shroom domain 2 reveals a three-segmented coiled-coil required for dimerization, Rock binding, and apical constriction.
Mohan, S., Rizaldy, R., Das, D., Bauer, R.J., Heroux, A., Trakselis, M.A., Hildebrand, J.D., VanDemark, A.P.(2012) Mol Biol Cell 23: 2131-2142
- PubMed: 22493320 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1091/mbc.E11-11-0937
- Primary Citation Related Structures:
3THF - PubMed Abstract:
Shroom (Shrm) proteins are essential regulators of cell shape and tissue morpho-logy during animal development that function by interacting directly with the coiled-coil region of Rho kinase (Rock). The Shrm-Rock interaction is sufficient to direct Rock subcellular localization and the subsequent assembly of contractile actomyosin networks in defined subcellular locales. However, it is unclear how the Shrm-Rock interaction is regulated at the molecular level. To begin investigating this issue, we present the structure of Shrm domain 2 (SD2), which mediates the interaction with Rock and is required for Shrm function. SD2 is a unique three-segmented dimer with internal symmetry, and we identify conserved residues on the surface and within the dimerization interface that are required for the Rock-Shrm interaction and Shrm activity in vivo. We further show that these residues are critical in both vertebrate and invertebrate Shroom proteins, indicating that the Shrm-Rock signaling module has been functionally and molecularly conserved. The structure and biochemical analysis of Shrm SD2 indicate that it is distinct from other Rock activators such as RhoA and establishes a new paradigm for the Rock-mediated assembly of contractile actomyosin networks.
- Department of Biological Sciences, University of Pittsburgh, Pittsburgh, PA 15260, USA.
Organizational Affiliation: