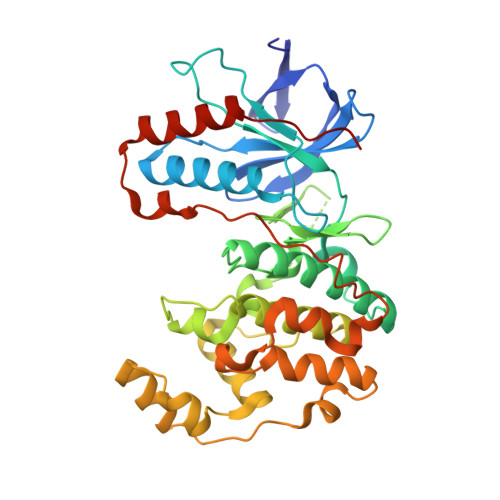
Resolving the selectivity problem for p38 mitogen activated protein (MAP) Kinase-inhibitors: Development of new highly potent inhibitors of p38 MAP kinase with an outstanding selectivity profile
Karcher, S.C., Fischer, S., Schattel, V., Romir, J., Stehle, T., Gruetter, C., Rauh, D., Laufer, S.A.To be published.