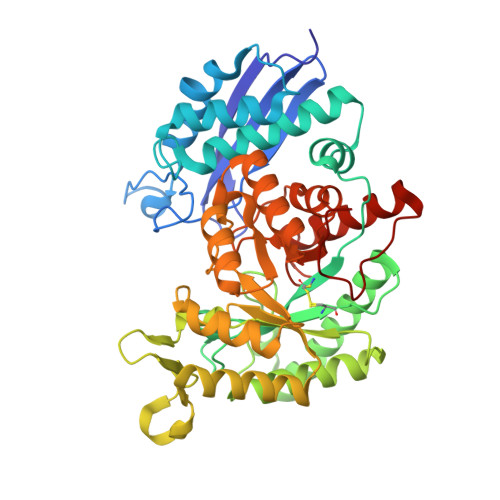
Structure analysis of Entamoeba histolytica enolase.
Schulz, E.C., Tietzel, M., Tovy, A., Ankri, S., Ficner, R.(2011) Acta Crystallogr D Biol Crystallogr 67: 619-627
- PubMed: 21697600 Search on PubMed
- DOI: https://doi.org/10.1107/S0907444911016544
- Primary Citation Related Structures:
3QTP - PubMed Abstract:
Entamoeba histolytica enolase (EhENO) reversibly interconverts 2-phosphoglyceric acid (2-PGA) and phosphoenolpyruvic acid (PEP). The crystal structure of the homodimeric EhENO is presented at a resolution of 1.9 Å. In the crystal structure EhENO presents as an asymmetric dimer with one active site in the open conformation and the other active site in the closed conformation. Interestingly, both active sites contain a copurified 2-PGA molecule. While the 2-PGA molecule in the closed active site closely resembles the conformation known from other enolase-2-PGA complexes, the conformation in the open active site is different. Here, 2-PGA is shifted approximately 1.6 Å away from metal ion I, most likely representing a precatalytic situation.
- Abteilung für Molekulare Strukturbiologie, Institut für Mikrobiologie und Genetik, Georg-August-Universität Göttingen, Justus-von-Liebig Weg, Germany. eschulz1@gwdg.de
Organizational Affiliation: