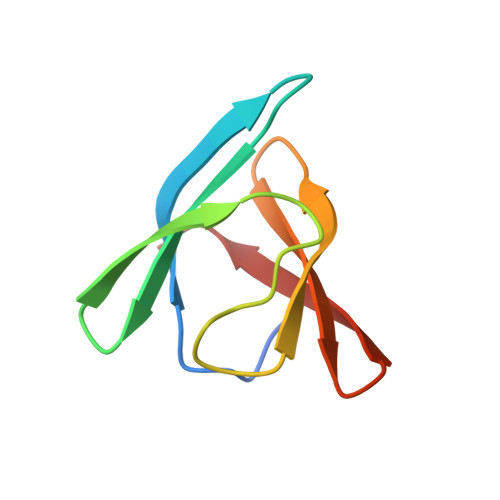
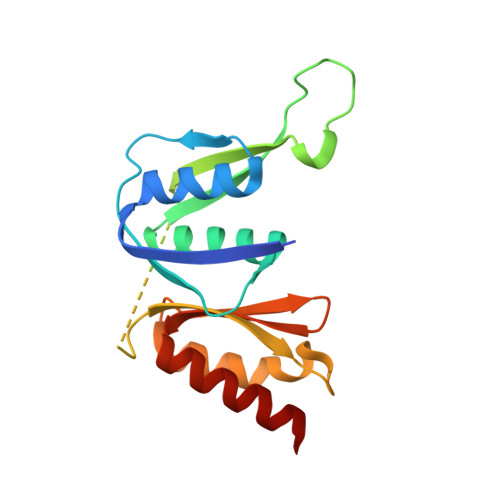
Structural and functional studies on the interaction of GspC and GspD in the type II secretion system.
Korotkov, K.V., Johnson, T.L., Jobling, M.G., Pruneda, J., Pardon, E., Heroux, A., Turley, S., Steyaert, J., Holmes, R.K., Sandkvist, M., Hol, W.G.(2011) PLoS Pathog 7: e1002228-e1002228
- PubMed: 21931548 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.ppat.1002228
- Primary Citation Related Structures:
3OSS - PubMed Abstract:
Type II secretion systems (T2SSs) are critical for secretion of many proteins from Gram-negative bacteria. In the T2SS, the outer membrane secretin GspD forms a multimeric pore for translocation of secreted proteins. GspD and the inner membrane protein GspC interact with each other via periplasmic domains. Three different crystal structures of the homology region domain of GspC (GspC(HR)) in complex with either two or three domains of the N-terminal region of GspD from enterotoxigenic Escherichia coli show that GspC(HR) adopts an all-β topology. N-terminal β-strands of GspC and the N0 domain of GspD are major components of the interface between these inner and outer membrane proteins from the T2SS. The biological relevance of the observed GspC-GspD interface is shown by analysis of variant proteins in two-hybrid studies and by the effect of mutations in homologous genes on extracellular secretion and subcellular distribution of GspC in Vibrio cholerae. Substitutions of interface residues of GspD have a dramatic effect on the focal distribution of GspC in V. cholerae. These studies indicate that the GspC(HR)-GspD(N0) interactions observed in the crystal structure are essential for T2SS function. Possible implications of our structures for the stoichiometry of the T2SS and exoprotein secretion are discussed.
- Department of Biochemistry, Biomolecular Structure Center, University of Washington, Seattle, Washington, USA.
Organizational Affiliation: