Crystal Structure of a Non-Neutralizing HIV-1 gp41 Envelope Antibody Demonstrates Neutralization Mechanism of gp41 Antibodies
Nicely, N.I., Dennison, S.M., Kelsoe, G., Ueda, Y., Liao, H.-X., Alam, S.M., Haynes, B.F.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| gp41 MPER-derived peptide | A [auth P] | 21 | Human immunodeficiency virus 1 | Mutation(s): 0 | 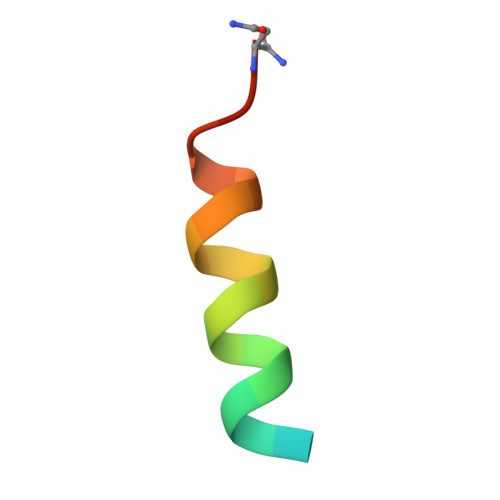 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P04578 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| ANTI-HIV-1 ANTIBODY 13H11 LIGHT CHAIN | B [auth A] | 224 | Mus musculus, Homo sapiens This entity is chimeric | Mutation(s): 0 | 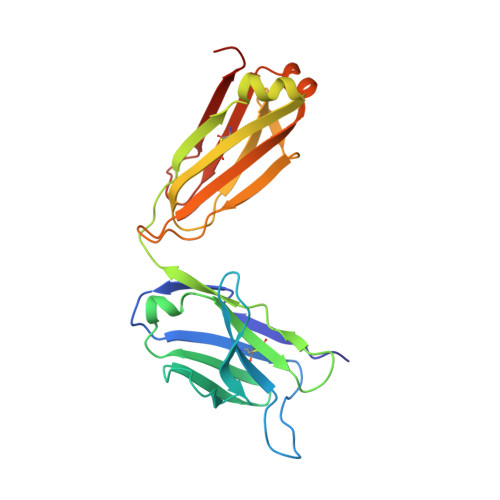 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q8TCD0 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| ANTI-HIV-1 ANTIBODY 13H11 HEAVY CHAIN | C [auth B] | 219 | Mus musculus, Homo sapiens This entity is chimeric | Mutation(s): 0 | 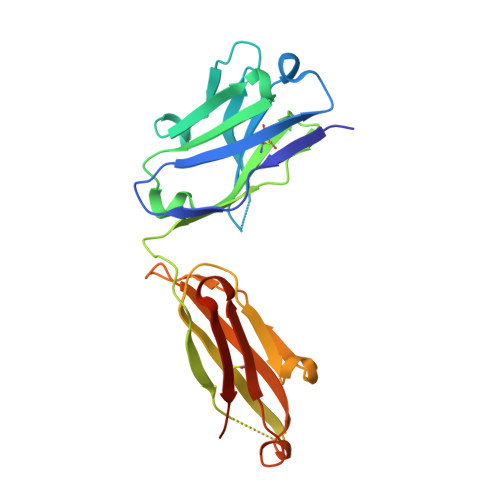 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | S6B291 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NA Download:Ideal Coordinates CCD File | D [auth B] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 66.89 | α = 90 |
| b = 121.692 | β = 90 |
| c = 58.097 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | model building |
| PHENIX | refinement |
| d*TREK | data reduction |
| d*TREK | data scaling |
| PHENIX | phasing |