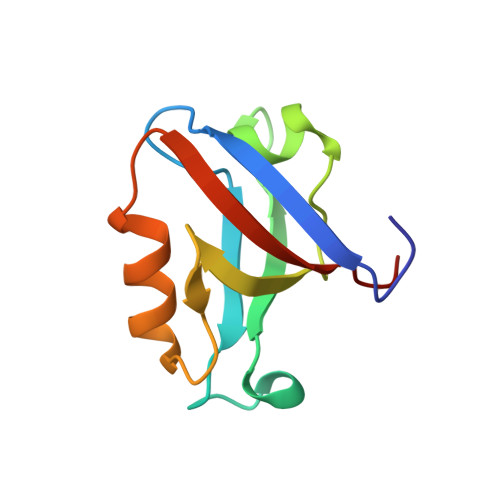
Crystallographic and nuclear magnetic resonance evaluation of the impact of peptide binding to the second PDZ domain of protein tyrosine phosphatase 1E.
Zhang, J., Sapienza, P.J., Ke, H., Chang, A., Hengel, S.R., Wang, H., Phillips, G.N., Lee, A.L.(2010) Biochemistry 49: 9280-9291
- PubMed: 20839809 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/bi101131f
- Primary Citation Related Structures:
3LNX, 3LNY - PubMed Abstract:
PDZ (PSD95/Discs large/ZO-1) domains are ubiquitous protein interaction motifs found in scaffolding proteins involved in signal transduction. Despite the fact that many PDZ domains show a limited tendency to undergo structural change, the PDZ family has been associated with long-range communication and allostery. One of the PDZ domains studied most in terms of structure and biophysical properties is the second PDZ ("PDZ2") domain from protein tyrosine phosphatase 1E (PTP1E, also known as PTPL1). Previously, we showed through NMR relaxation studies that binding of the RA-GEF2 C-terminal peptide substrate results in long-range propagation of side-chain dynamic changes in human PDZ2 [Fuentes, E. J., et al. (2004) J. Mol. Biol. 335, 1105-1115]. Here, we present the first X-ray crystal structures of PDZ2 in the absence and presence of RA-GEF2 ligand, determined to resolutions of 1.65 and 1.3 Å, respectively. These structures deviate somewhat from previously determined NMR structures and indicate that very minor structural changes in PDZ2 accompany peptide binding. NMR residual dipolar couplings confirm the crystal structures to be accurate models of the time-averaged atomic coordinates of PDZ2. The impact on side-chain dynamics was further tested with a C-terminal peptide from APC, which showed results nearly identical to those of RA-GEF2. Thus, allosteric transmission in PDZ2 induced by peptide binding is conveyed purely and robustly by dynamics. (15)N relaxation dispersion measurements did not detect appreciable populations of a kinetic structural intermediate. Collectively, for ligand binding to PDZ2, these data support a lock-and-key binding model from a structural perspective and an allosteric model from a dynamical perspective, which together suggest a complex energy landscape for functional transitions within the ensemble.
- Department of Biochemistry and Biophysics, School of Medicine, University of North Carolina, Chapel Hill, NC 27599, USA.
Organizational Affiliation: