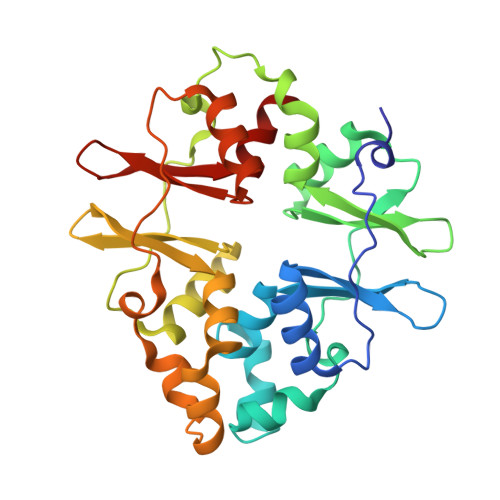
The crystal structure of protein MJ1225 from Methanocaldococcus jannaschii shows strong conservation of key structural features seen in the eukaryal gamma-AMPK.
Gomez-Garcia, I., Oyenarte, I., Martinez-Cruz, L.A.(2010) J Mol Biology 65: 813-817
- PubMed: 20382158 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2010.03.045
- Primary Citation Related Structures:
3KH5, 3LFZ - PubMed Abstract:
In mammals, 5'-AMP-activated protein kinase (AMPK) is a heterotrimeric protein composed of a catalytic serine/threonine kinase subunit (alpha) and two regulatory subunits (beta and gamma). The gamma-subunit senses the intracellular energy status by competitively binding AMP and ATP and is thought to be responsible for allosteric regulation of the whole complex. We describe herein the crystal structure of protein MJ1225 from Methanocaldococcus jannaschii complexed to AMP, ADP, and ATP. Our data provide evidence of a strong conservation of the key functional features seen in the gamma-subunit of the eukaryotic AMPK, and more importantly, it reveals a novel AMP binding site, herein denoted as site E, which had not been previously described in cystathionine beta-synthase domains so far. Site E is located in a small cavity existing between the alpha-helices structurally equivalent to those disrupting the internal symmetry of each Bateman domain in gamma-AMPKs and shows striking similarities with a symmetry-related crevice of the mammalian enzyme that hosts the pathological mutation N488I.
- Structural Biology Unit, CIC bioGUNE, Parque Tecnológico de Bizkaia, Edificio 800, 48160 Derio, Bizkaia, Spain.
Organizational Affiliation: