Molecular Links between the E2 Envelope Glycoprotein and Nucleocapsid Core in Sindbis Virus.
Tang, J., Jose, J., Chipman, P., Zhang, W., Kuhn, R.J., Baker, T.S.(2011) J Mol Biology 414: 442-459
- PubMed: 22001018 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jmb.2011.09.045
- Primary Citation Related Structures:
3J0F - PubMed Abstract:
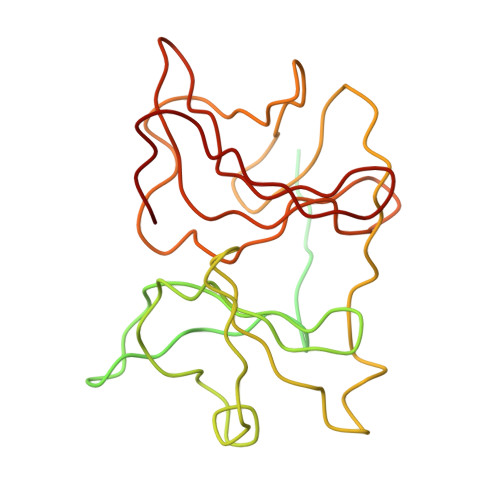
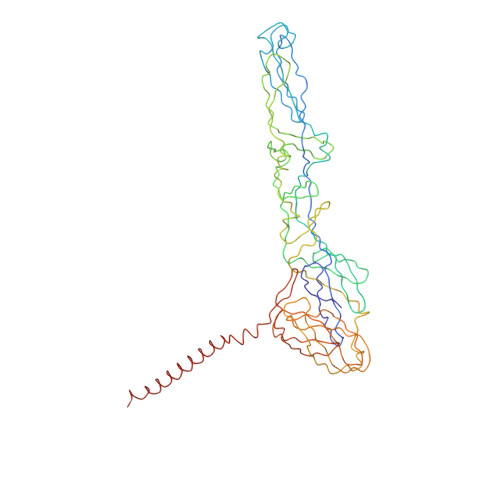
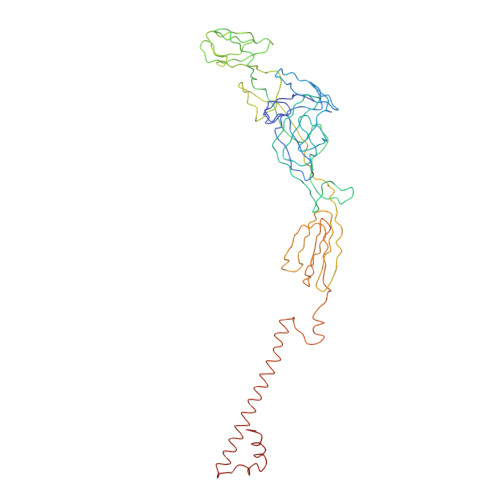
A three-dimensional reconstruction of Sindbis virus at 7.0 Å resolution presented here provides a detailed view of the virion structure and includes structural evidence for key interactions that occur between the capsid protein (CP) and transmembrane (TM) glycoproteins E1 and E2. Based on crystal structures of component proteins and homology modeling, we constructed a nearly complete, pseudo-atomic model of the virus. Notably, this includes identification of the 33-residue cytoplasmic domain of E2 (cdE2), which follows a path from the E2 TM helix to the CP where it enters and exits the CP hydrophobic pocket and then folds back to contact the viral membrane. Modeling analysis identified three major contact regions between cdE2 and CP, and the roles of specific residues were probed by molecular genetics. This identified R393 and E395 of cdE2 and Y162 and K252 of CP as critical for virus assembly. The N-termini of the CPs form a contiguous network that interconnects 12 pentameric and 30 hexameric CP capsomers. A single glycoprotein spike cross-links three neighboring CP capsomers as might occur during initiation of virus budding.
- Department of Chemistry and Biochemistry, University of California, San Diego, La Jolla, CA 92093-0378, USA.
Organizational Affiliation: