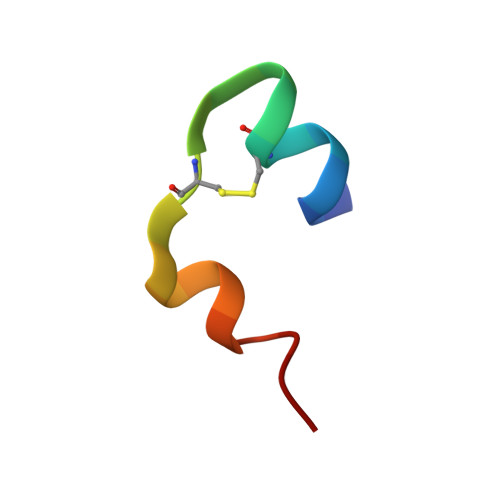
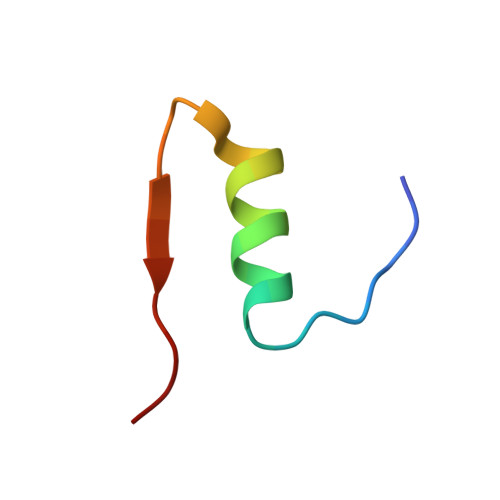
Structure of insulin: results of joint neutron and X-ray refinement.
Wlodawer, A., Savage, H., Dodson, G.(1989) Acta Crystallogr B 45: 99-107
- PubMed: 2695122 Search on PubMed
- DOI: https://doi.org/10.1107/s0108768188011012
- Primary Citation Related Structures:
3INS - PubMed Abstract:
Neutron diffraction data for porcine 2Zn insulin were collected to 2.2 A resolution from a single crystal deuterated by slow exchange of mother liquor. A joint neutron/X-ray restrained-least-squares refinement was undertaken using the neutron data, as well as the 1.5 A resolution X-ray data collected previously. The final R factors were 0.182 for the X-ray data and 0.191 for the neutron data. Resulting atomic coordinates were compared with the initial X-ray model, showing a total r.m.s. shift of 0.36 A for the protein and 0.6 A for the solvent. Protonation of a number of individual amino acids was investigated by analysis of the neutron maps. No D atoms were found between the carboxylates of Glu B13 which make an intermolecular contact, suggesting nonbonded interaction rather than the predicted hydrogen bond. Amide hydrogen exchange was investigated in a refinement of their atomic occupancies. Regions of unexchanged amide groups were found in the center of the B helices. The results of this study emphasize the limited amount of information available in neutron diffraction studies of proteins at resolution lower than 2 A.
- Center for Chemical Physics, National Bureau of Standards, Gaithersburg, MD 20899.
Organizational Affiliation: