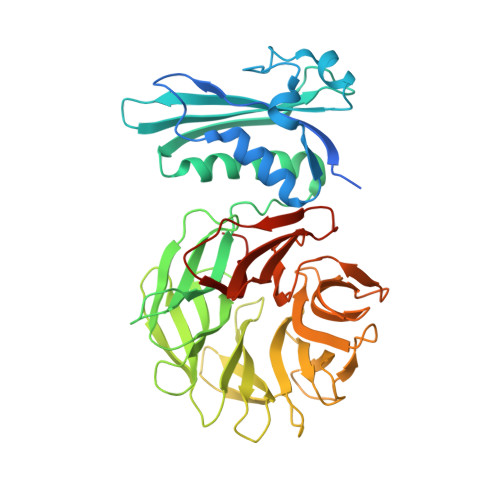
The crystal structure of the TolB box of colicin A in complex with TolB reveals important differences in the recruitment of the common TolB translocation portal used by group A colicins.
Zhang, Y., Li, C., Vankemmelbeke, M.N., Bardelang, P., Paoli, M., Penfold, C.N., James, R.(2009) Mol Microbiol 75: 623-636
- PubMed: 19627502 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1111/j.1365-2958.2009.06808.x
- Primary Citation Related Structures:
3IAX - PubMed Abstract:
Interaction of the TolB box of Group A colicins with the TolB protein in the periplasm of Escherichia coli cells promotes transport of the cytotoxic domain of the colicin across the cell envelope. The crystal structure of a complex between a 107-residue peptide (TA(1-107)) of the translocation domain of colicin A (ColA) and TolB identified the TolB box as a 12-residue peptide that folded into a distorted hairpin within a central canyon of the beta-propeller domain of TolB. Comparison of this structure with that of the colicin E9 (ColE9) TolB box-TolB complex, together with site-directed mutagenesis of the ColA TolB box residues, revealed important differences in the interaction of the two TolB boxes with an overlapping binding site on TolB. Substitution of the TolB box residues of ColA with those of ColE9 conferred the ability to competitively recruit TolB from Pal but reduced the biological activity of the mutant ColA. This datum explains (i) the difference in binding affinities of ColA and ColE9 with TolB, and (ii) the inability of ColA, unlike ColE9, to competitively recruit TolB from Pal, allowing an understanding of how these two colicins interact in a different way with a common translocation portal in E. coli cells.
- Institute of Infection, Immunity and Inflammation, School of Molecular Medical Sciences, University of Nottingham, University Park, Nottingham NG7 2RD, UK.
Organizational Affiliation: