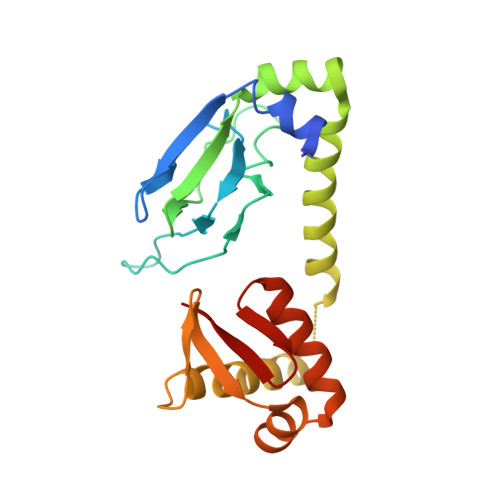
Structure of apo-CAP reveals that large conformational changes are necessary for DNA binding.
Sharma, H., Yu, S., Kong, J., Wang, J., Steitz, T.A.(2009) Proc Natl Acad Sci U S A 106: 16604-16609
- PubMed: 19805344 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0908380106
- Primary Citation Related Structures:
3FWE, 3HIF - PubMed Abstract:
The binding of cAMP to the Escherichia coli catabolite gene activator protein (CAP) produces a conformational change that enables it to bind specific DNA sequences and regulate transcription, which it cannot do in the absence of the nucleotide. The crystal structures of the unliganded CAP containing a D138L mutation and the unliganded WT CAP were determined at 2.3 and 3.6 A resolution, respectively, and reveal that the two DNA binding domains have dimerized into one rigid body and their two DNA recognition helices become buried. The WT structure shows multiple orientations of this rigid body relative to the nucleotide binding domain supporting earlier biochemical data suggesting that the inactive form exists in an equilibrium among different conformations. Comparison of the structures of the liganded and unliganded CAP suggests that cAMP stabilizes the active DNA binding conformation of CAP through the interactions that the N(6) of the adenosine makes with the C-helices. These interactions are associated with the reorientation and elongation of the C-helices that precludes the formation of the inactive structure.
- Departments of Chemistry and Molecular Biophysics and Biochemistry, Yale University, Howard Hughes Medical Institute, New Haven, CT 06511, USA.
Organizational Affiliation: