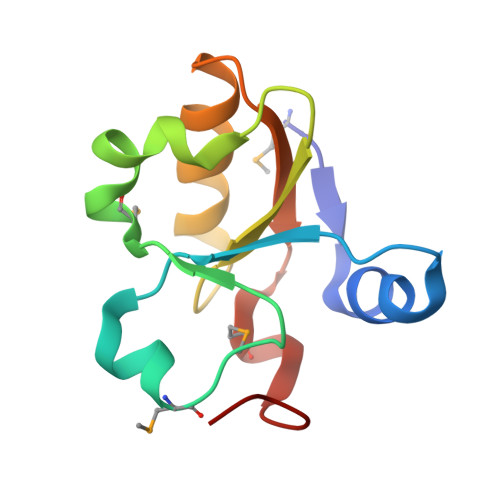
Crystal Structure of SSP1007 From Staphylococcus saprophyticus subsp. saprophyticus. Northeast Structural Genomics Target SyR101A.
Seetharaman, J., Abashidze, M., Wang, H., Janjua, H., Foote, E.L., Xiao, R., Everett, J.K., Acton, T.B., Rost, B., Montelione, G.T., Hunt, J.F., Tong, L.To be published.