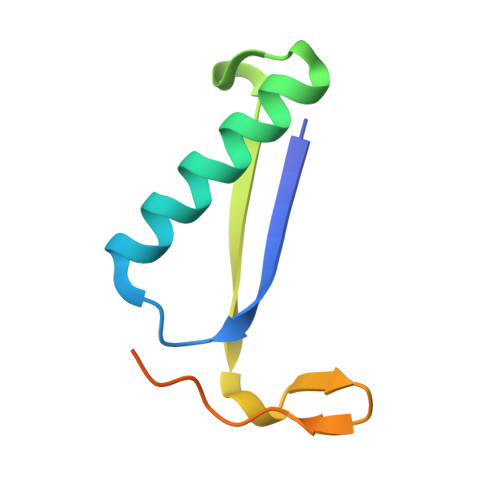
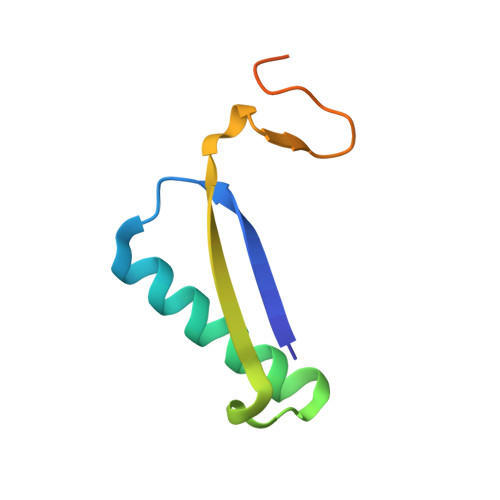
Structural and mechanistic analysis of trans-3-chloroacrylic acid dehalogenase activity.
Pegan, S.D., Serrano, H., Whitman, C.P., Mesecar, A.D.(2008) Acta Crystallogr D Biol Crystallogr 64: 1277-1282
- PubMed: 19018104
- DOI: https://doi.org/10.1107/S0907444908034707
- Primary Citation Related Structures:
3EJ3, 3EJ7, 3EJ9 - PubMed Abstract:
Trans-3-chloroacrylic acid dehalogenase (CaaD) is a critical enzyme in the trans-1,3-dichloropropene (DCP) degradation pathway in Pseudomonas pavonaceae 170. This enzyme allows bacteria to use trans-DCP, a common component in commercially produced fumigants, as a carbon source. CaaD specifically catalyzes the fourth step of the pathway by cofactor-independent dehalogenation of a vinyl carbon-halogen bond. Previous studies have reported an X-ray structure of CaaD under acidic conditions with a covalent modification of the catalytic betaPro1 residue. Here, the 1.7 A resolution X-ray structure of CaaD under neutral (pH 6.5) conditions is reported without the presence of the covalent adduct. In this new structure, a substrate-like acetate molecule is bound within the active site in a position analogous to the putative substrate-binding site. Additionally, a catalytically important water molecule was identified, consistent with previously proposed reaction schemes. Finally, flexibility of the catalytically relevant side chain alphaGlu52 is observed in the structure, supporting its role in the catalytic mechanism.
- Center of Pharmaceutical Biotechnology and Department of Medicinal Chemistry and Pharmacognosy, University of Illinois, Chicago, USA. pegan@uic.edu
Organizational Affiliation: