The crystal structure of Tudor domain of human Histone-lysine N-methyltransferase SETDB1.
Dombrovski, L., Amaya, M.F., Loppnau, P., Bochkarev, A., Min, J., Wu, H.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Histone-lysine N-methyltransferase SETDB1 | 213 | Homo sapiens | Mutation(s): 0 Gene Names: SETDB1, KIAA0067, KMT1E EC: 2.1.1.43 (PDB Primary Data), 2.1.1.366 (UniProt) | 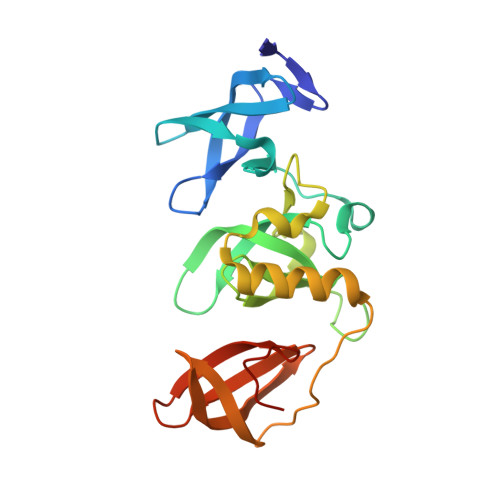 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q15047 GTEx: ENSG00000143379 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q15047 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 54.518 | α = 90 |
| b = 63.689 | β = 90 |
| c = 69.083 | γ = 90 |
| Software Name | Purpose |
|---|---|
| DENZO | data reduction |
| SCALEPACK | data scaling |
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |
| SHELXD | phasing |