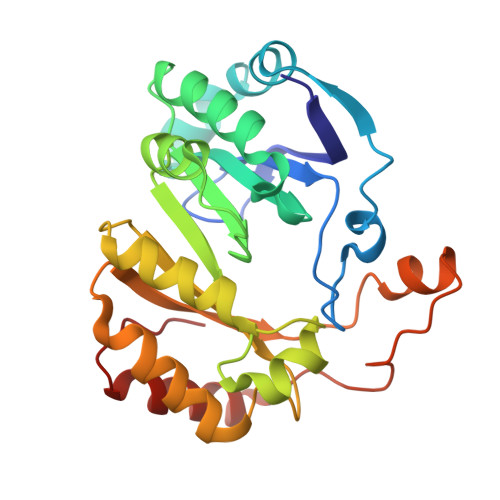
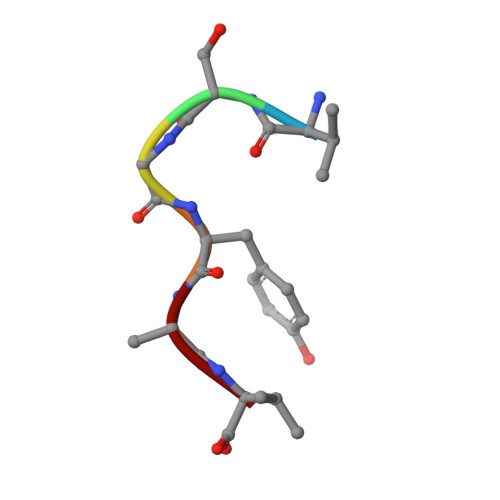
Structural basis for glycyl radical formation by pyruvate formate-lyase activating enzyme.
Vey, J.L., Yang, J., Li, M., Broderick, W.E., Broderick, J.B., Drennan, C.L.(2008) Proc Natl Acad Sci U S A 105: 16137-16141
- PubMed: 18852451 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0806640105
- Primary Citation Related Structures:
3C8F, 3CB8 - PubMed Abstract:
Pyruvate formate-lyase activating enzyme generates a stable and catalytically essential glycyl radical on G(734) of pyruvate formate-lyase via the direct, stereospecific abstraction of a hydrogen atom from pyruvate formate-lyase. The activase performs this remarkable feat by using an iron-sulfur cluster and S-adenosylmethionine (AdoMet), thus placing it among the AdoMet radical superfamily of enzymes. We report here structures of the substrate-free and substrate-bound forms of pyruvate formate-lyase-activating enzyme, the first structures of an AdoMet radical activase. To obtain the substrate-bound structure, we have used a peptide substrate, the 7-mer RVSGYAV, which contains the sequence surrounding G(734). Our structures provide fundamental insights into the interactions between the activase and the G(734) loop of pyruvate formate-lyase and provide a structural basis for direct and stereospecific H atom abstraction from the buried G(734) of pyruvate formate-lyase.
- Departments of Chemistry and Biology, Massachusetts Institute of Technology, Cambridge, MA 02139, USA.
Organizational Affiliation: