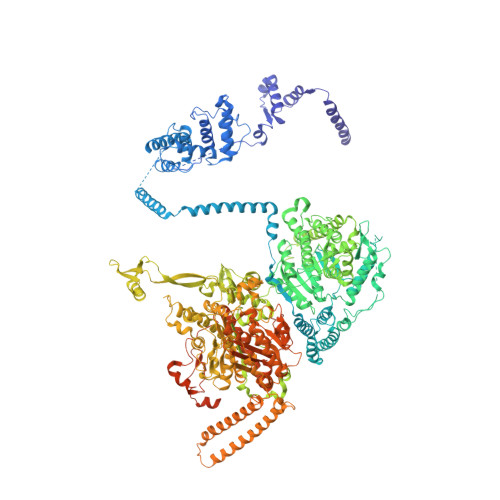
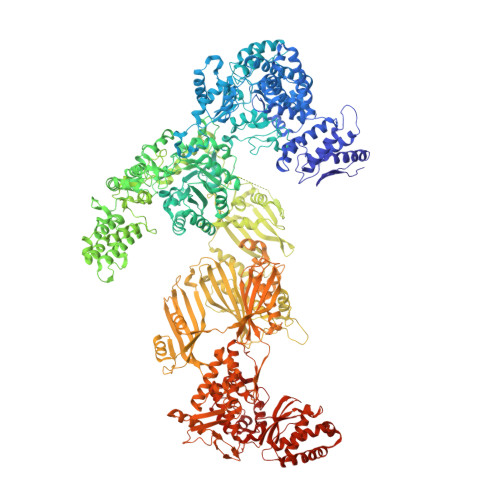
Inhibition of the Fungal Fatty Acid Synthase Type I Multienzyme Complex.
Johansson, P., Wiltschi, B., Kumari, P., Kessler, B., Vonrhein, C., Vonck, J., Oesterhelt, D., Grininger, M.(2008) Proc Natl Acad Sci U S A 105: 12803
- PubMed: 18725634 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0805827105
- Primary Citation Related Structures:
2VKZ - PubMed Abstract:
Fatty acids are among the major building blocks of living cells, making lipid biosynthesis a potent target for compounds with antibiotic or antineoplastic properties. We present the crystal structure of the 2.6-MDa Saccharomyces cerevisiae fatty acid synthase (FAS) multienzyme in complex with the antibiotic cerulenin, representing, to our knowledge, the first structure of an inhibited fatty acid megasynthase. Cerulenin attacks the FAS ketoacyl synthase (KS) domain, forming a covalent bond to the active site cysteine C1305. The inhibitor binding causes two significant conformational changes of the enzyme. First, phenylalanine F1646, shielding the active site, flips and allows access to the nucleophilic cysteine. Second, methionine M1251, placed in the center of the acyl-binding tunnel, rotates and unlocks the inner part of the fatty acid binding cavity. The importance of the rotational movement of the gatekeeping M1251 side chain is reflected by the cerulenin resistance and the changed product spectrum reported for S. cerevisiae strains mutated in the adjacent glycine G1250. Platensimycin and thiolactomycin are two other potent inhibitors of KSs. However, in contrast to cerulenin, they show selectivity toward the prokaryotic FAS system. Because the flipped F1646 characterizes the catalytic state accessible for platensimycin and thiolactomycin binding, we superimposed structures of inhibited bacterial enzymes onto the S. cerevisiae FAS model. Although almost all side chains involved in inhibitor binding are conserved in the FAS multienzyme, a different conformation of the loop K1413-K1423 of the KS domain might explain the observed low antifungal properties of platensimycin and thiolactomycin.
- Department of Membrane Biochemistry, Max Planck Institute of Biochemistry, Am Klopferspitz 18, 82152 Martinsried, Germany.
Organizational Affiliation: