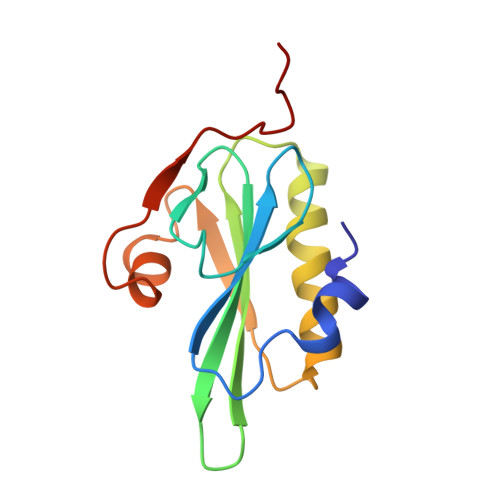
Refined structure of villin 14T and a detailed comparison with other actin-severing domains.
Markus, M.A., Matsudaira, P., Wagner, G.(1997) Protein Sci 6: 1197-1209
- PubMed: 9194180 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1002/pro.5560060608
- Primary Citation Related Structures:
2VIK, 2VIL - PubMed Abstract:
Villin 14T is the amino terminal actin monomer binding domain from the actin-severing and bundling protein villin. Its structure has been determined in solution using heteronuclear multidimensional nuclear magnetic resonance (NMR) spectroscopy (Markus MA, Nakayama T, Matsudaira P, Wagner G. 1994. Solution structure of villin 14T, a domain conserved among actin-severing proteins. Protein Science 3:70-81). An additional nuclear Overhauser effect (NOE) spectroscopy data set, acquired using improved gradient techniques, and further detailed analysis of existing data sets, produced an additional 601 NOE restraints for structure calculations. The overall fold does not change significantly with the additional NOE restraints but the definition of the structure is improved, as judged by smaller deviations among an ensemble of calculated structures that adequately satisfy the NMR restraints. Some of the side chains, especially those in the hydrophobic core of the domain, are much more defined. This improvement in the detail of the solution structure of villin 14T makes it interesting to compare the structure with the crystal structure of gelsolin segment 1, which shares 58% sequence identity with villin 14T, in an effort to gain insight into villin 14T's weaker affinity for actin monomers. Villin 14T has smaller side chains at several positions that make hydrophobic contacts with actin in the context of gelsolin segment 1. The structure is also compared with the structure of the related actin-severing domain, severin domain 2.
- Committee on Higher Degrees in Biophysics, Harvard University, Boston, Massachusetts 02115, USA.
Organizational Affiliation: