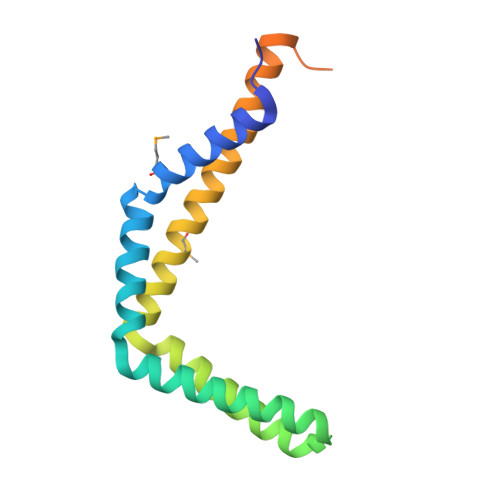
Crystal structure of Protein ymcA from Bacillus subtilis.
Seetharaman, J., Abashidze, M., Forouhar, F., Wang, D., Fang, Y., Cunningham, K., Ma, L.-C., Xia, R., Liu, J., Baran, M.C., Acton, T.B., Rost, B., Montelione, G.T., Tong, L., Hunt, J.F.To be published.