The structure of probable glutaminase from B. subtilis complexed with its inhibitor 6-diazo-5-oxo-L-norleucine
Kim, Y., Dementieva, I., Vinokour, E., Collart, F., Joachimiak, A.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Glutaminase 1 | 330 | Bacillus subtilis | Mutation(s): 12 Gene Names: glsA1, glsA, BSU02430 EC: 3.5.1.2 | 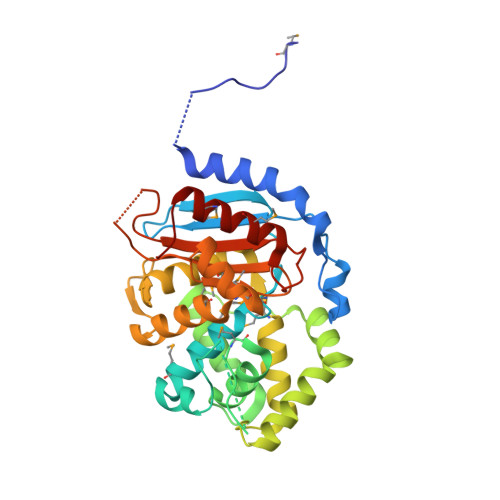 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O31465 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| DON Download:Ideal Coordinates CCD File | C [auth A], D [auth B] | 6-DIAZENYL-5-OXO-L-NORLEUCINE C6 H11 N3 O3 CKRLZVNSTCQAJW-YFKPBYRVSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A, B | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 71.247 | α = 90 |
| b = 184.701 | β = 90 |
| c = 51.422 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| SBC-Collect | data collection |
| HKL-2000 | data collection |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| HKL-3000 | phasing |