New Antimicrobial Potential and Structural Properties of PAFB: A Cationic, Cysteine-Rich Protein from Penicillium chrysogenum Q176.
Huber, A., Hajdu, D., Bratschun-Khan, D., Gaspari, Z., Varbanov, M., Philippot, S., Fizil, A., Czajlik, A., Kele, Z., Sonderegger, C., Galgoczy, L., Bodor, A., Marx, F., Batta, G.(2018) Sci Rep 8: 1751-1751
- PubMed: 29379111
- DOI: https://doi.org/10.1038/s41598-018-20002-2
- Primary Citation of Related Structures:
2NC2 - PubMed Abstract:
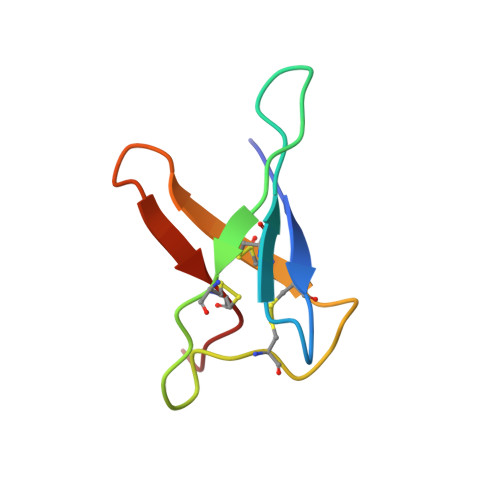
Small, cysteine-rich and cationic proteins with antimicrobial activity are produced by diverse organisms of all kingdoms and represent promising molecules for drug development. The ancestor of all industrial penicillin producing strains, the ascomycete Penicillium chryosgenum Q176, secretes the extensively studied antifungal protein PAF. However, the genome of this strain harbours at least two more genes that code for other small, cysteine-rich and cationic proteins with potential antifungal activity. In this study, we characterized the pafB gene product that shows high similarity to PgAFP from P. chrysogenum R42C. Although abundant and timely regulated pafB gene transcripts were detected, we could not identify PAFB in the culture broth of P. chrysogenum Q176. Therefore, we applied a P. chrysogenum-based expression system to produce sufficient amounts of recombinant PAFB to address unanswered questions concerning the structure and antimicrobial function. Nuclear magnetic resonance (NMR)-based analyses revealed a compact β-folded structure, comprising five β-strands connected by four solvent exposed and flexible loops and an "abcabc" disulphide bond pattern. We identified PAFB as an inhibitor of growth of human pathogenic moulds and yeasts. Furthermore, we document for the first time an anti-viral activity for two members of the small, cysteine-rich and cationic protein group from ascomycetes.
- Division of Molecular Biology, Biocenter, Medical University of Innsbruck, Innrain 80-82, 6020, Innsbruck, Austria.
Organizational Affiliation: