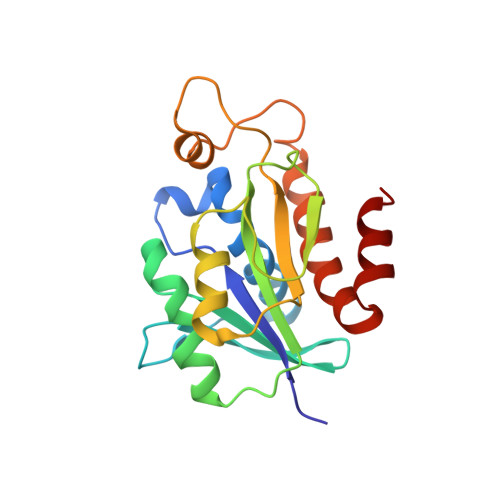
Structural characterization of peptidyl-tRNA hydrolase from Mycobacterium smegmatis by NMR spectroscopy.
Kabra, A., Fatma, F., Shahid, S., Pathak, P.P., Yadav, R., Pulavarti, S.V., Tripathi, S., Jain, A., Arora, A.(2016) Biochim Biophys Acta 1864: 1304-1314
- PubMed: 27378575
- DOI: https://doi.org/10.1016/j.bbapap.2016.06.013
- Primary Citation Related Structures:
2NAF - PubMed Abstract:
Accumulation of toxic peptidyl-tRNAs in the bacterial cytoplasm is averted by the action of peptidyl-tRNA hydrolase (Pth), which cleaves peptidyl-tRNA into free tRNA and peptide. NMR studies are needed for a protein homolog with a complete crystal structure, for comparison with the NMR structure of Mycobacterium tuberculosis Pth. The structure and dynamics of Mycobacterium smegmatis Pth (MsPth) were characterized by NMR spectroscopy and MD simulations. The thermal stability of MsPth was characterized by DSC. MsPth NMR structure has a central mixed seven stranded β-sheet that is enclosed by six α-helices. NMR relaxation and MD simulations studies show that most of the ordered regions are rigid. Of the substrate binding segments, the gate loop is rigid, the base loop displays slow motions, while the lid loop displays fast timescale motions. MsPth displays high thermal stability characterized by a melting temperature of 61.71°C. The NMR structure of MsPth shares the canonical Pth fold with the NMR structure of MtPth. The motional characteristics for the lid region, the tip of helix α3, and the gate region, as indicated by MD simulations and NMR data, are similar for MsPth and MtPth. However, MsPth has relatively less rigid base loop and more compactly packed helices α5 and α6. The packing and the dynamic differences appear to be an important contributing factor to the thermal stability of MsPth, which is significantly higher than that of MtPth. MsPth structure consolidates our understanding of the structure and dynamics of bacterial Pth proteins.
- Molecular and Structural Biology Division, CSIR-Central Drug Research Institute, Lucknow 226031, India.
Organizational Affiliation: