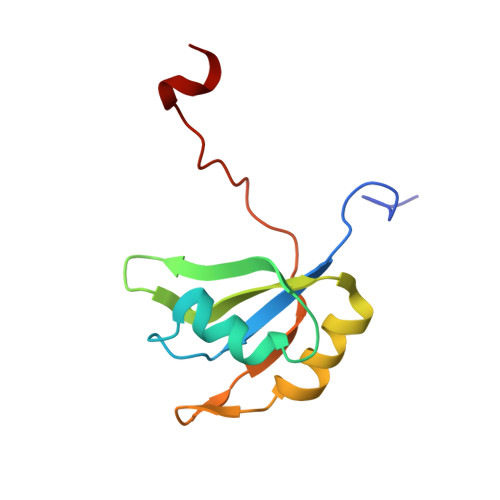
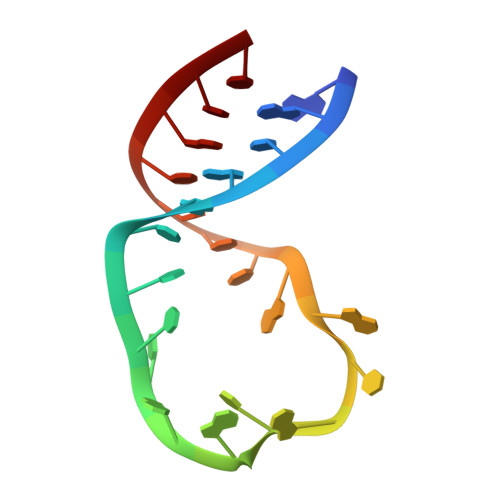
Rbfox proteins regulate microRNA biogenesis by sequence-specific binding to their precursors and target downstream Dicer.
Chen, Y., Zubovic, L., Yang, F., Godin, K., Pavelitz, T., Castellanos, J., Macchi, P., Varani, G.(2016) Nucleic Acids Res 44: 4381-4395
- PubMed: 27001519 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkw177
- Primary Citation Related Structures:
2N7X, 2N82 - PubMed Abstract:
Rbfox proteins regulate tissue-specific splicing by targeting a conserved GCAUG sequence within pre-mRNAs. We report here that sequence-specific binding of the conserved Rbfox RRM to miRNA precursors containing the same sequence motif in their terminal loops, including miR-20b and miR-107, suppresses their nuclear processing. The structure of the complex between precursor miR-20b and Rbfox RRM shows the molecular basis for recognition, and reveals changes in the stem-loop upon protein binding. In mammalian cells, Rbfox2 downregulates mature miR-20b and miR-107 levels and increases the expression of their downstream targets PTEN and Dicer, respectively, suggesting that Rbfox2 indirectly regulates many more cellular miRNAs. Thus, some of the widespread cellular functions of Rbfox2 protein are attributable to regulation of miRNA biogenesis, and might include the mis-regulation of miR-20b and miR-107 in cancer and neurodegeneration.
- Center for Integrative Biology (CIBIO), University of Trento, Via Sommarive 9, 38123 Trento (TN), Italy.
Organizational Affiliation: