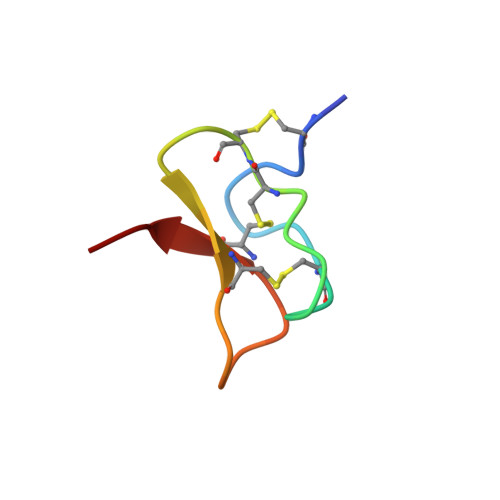
Identification and Characterization of ProTx-III [ mu-TRTX-Tp1a], a New Voltage-Gated Sodium Channel Inhibitor from Venom of the Tarantula Thrixopelma pruriens.
Cardoso, F.C., Dekan, Z., Rosengren, K.J., Erickson, A., Vetter, I., Deuis, J.R., Herzig, V., Alewood, P.F., King, G.F., Lewis, R.J.(2015) Mol Pharmacol 88: 291-303
- PubMed: 25979003 Search on PubMed
- DOI: https://doi.org/10.1124/mol.115.098178
- Primary Citation Related Structures:
2MXM - PubMed Abstract:
Spider venoms are a rich source of ion channel modulators with therapeutic potential. Given the analgesic potential of subtype-selective inhibitors of voltage-gated sodium (NaV) channels, we screened spider venoms for inhibitors of human NaV1.7 (hNaV1.7) using a high-throughput fluorescent assay. Here, we describe the discovery of a novel NaV1.7 inhibitor, μ-TRTX-Tp1a (Tp1a), isolated from the venom of the Peruvian green-velvet tarantula Thrixopelma pruriens. Recombinant and synthetic forms of this 33-residue peptide preferentially inhibited hNaV1.7 > hNaV1.6 > hNaV1.2 > hNaV1.1 > hNaV1.3 channels in fluorescent assays. NaV1.7 inhibition was diminished (IC50 11.5 nM) and the association rate decreased for the C-terminal acid form of Tp1a compared with the native amidated form (IC50 2.1 nM), suggesting that the peptide C terminus contributes to its interaction with hNaV1.7. Tp1a had no effect on human voltage-gated calcium channels or nicotinic acetylcholine receptors at 5 μM. Unlike most spider toxins that modulate NaV channels, Tp1a inhibited hNaV1.7 without significantly altering the voltage dependence of activation or inactivation. Tp1a proved to be analgesic by reversing spontaneous pain induced in mice by intraplantar injection in OD1, a scorpion toxin that potentiates hNaV1.7. The structure of Tp1a as determined using NMR spectroscopy revealed a classic inhibitor cystine knot (ICK) motif. The molecular surface of Tp1a presents a hydrophobic patch surrounded by positively charged residues, with subtle differences from other ICK spider toxins that might contribute to its different pharmacological profile. Tp1a may help guide the development of more selective and potent hNaV1.7 inhibitors for treatment of chronic pain.
- Institute for Molecular Bioscience (F.C.C., Z.D., I.V., J.R.D., V.H., P.F.A., G.F.K., R.J.L.), School of Biomedical Sciences (K.J.R.), and School of Chemistry and Molecular Biosciences (A.E.), The University of Queensland, Brisbane, Queensland, Australia.
Organizational Affiliation: