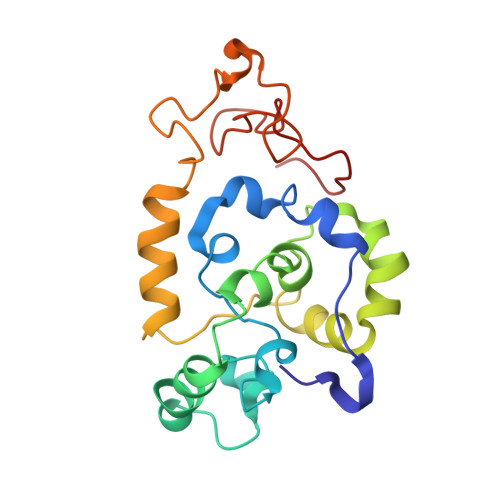
Spider wrapping silk fibre architecture arising from its modular soluble protein precursor.
Tremblay, M.L., Xu, L., Lefevre, T., Sarker, M., Orrell, K.E., Leclerc, J., Meng, Q., Pezolet, M., Auger, M., Liu, X.Q., Rainey, J.K.(2015) Sci Rep 5: 11502-11502
- PubMed: 26112753
- DOI: https://doi.org/10.1038/srep11502
- Primary Citation Related Structures:
2MU3 - PubMed Abstract:
Spiders store spidroins in their silk glands as high concentration aqueous solutions, spinning these dopes into fibres with outstanding mechanical properties. Aciniform (or wrapping) silk is the toughest spider silk and is devoid of the short amino acid sequence motifs characteristic of the other spidroins. Using solution-state NMR spectroscopy, we demonstrate that the 200 amino acid Argiope trifasciata AcSp1 repeat unit contrasts with previously characterized spidroins, adopting a globular 5-helix bundle flanked by intrinsically disordered N- and C-terminal tails. Split-intein-mediated segmental NMR-active isotope-enrichment allowed unambiguous demonstration of modular and malleable "beads-on-a-string" concatemeric behaviour. Concatemers form fibres upon manual drawing with silk-like morphology and mechanical properties, alongside secondary structuring and orientation consistent with native AcSp1 fibres. AcSp1 structural stability varies locally, with the fifth helix denaturing most readily. The structural transition of aciniform spidroin from a mostly α-helical dope to a mixed α-helix/β-sheet-containing fibre can be directly related to spidroin architecture and stability.
- Department of Biochemistry &Molecular Biology, Dalhousie University, Halifax, NS, Canada.
Organizational Affiliation: