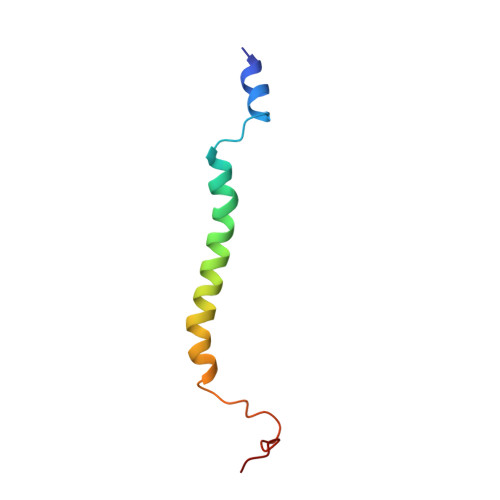
Solution structure of the transmembrane domain of the insulin receptor in detergent micelles
Li, Q., Wong, Y.L., Kang, C.(2014) Biochim Biophys Acta 1838: 1313-1321
- PubMed: 24440425 Search on PubMed
- DOI: https://doi.org/10.1016/j.bbamem.2014.01.005
- Primary Citation Related Structures:
2MFR - PubMed Abstract:
The insulin receptor (IR) binds insulin and plays important roles in glucose homeostasis by regulating the tyrosine kinase activity at its C-terminus. Its transmembrane domain (TMD) is shown to be important for transferring conformational changes induced by insulin across the cell membrane to regulate kinase activity. In this study, a construct IR(940-988) containing the TMD was expressed and purified for structural studies. Its solution structure in dodecylphosphocholine (DPC) micelles was determined. The sequence containing residues L962 to Y976 of the TMD of the IR in micelles adopts a well-defined helical structure with a kink formed by glycine and proline residues present at its N-terminus, which might be important for its function. Paramagnetic relaxation enhancement (PRE) and relaxation experimental results suggest that residues following the TMD are flexible and expose to aqueous solution. Although purified IR(940-988) in micelles existed mainly as a monomeric form verified by gel filtration and relaxation analysis, cross-linking study suggests that it may form a dimer or oligomers under micelle conditions.
- Institute of Chemical & Engineering Sciences, Agency for Science, Technology and Research (A*STAR), Singapore, Singapore.
Organizational Affiliation: