Solution Structure of MSL2 CXC Domain Reveals an Unusual Zn(3)Cys(9) Cluster and Similarity to Pre-SET Domains of Histone Lysine Methyltransferases.
Zheng, S., Wang, J., Feng, Y., Wang, J., Ye, K.(2012) PLoS One 7: e45437-e45437
- PubMed: 23029009
- DOI: https://doi.org/10.1371/journal.pone.0045437
- Primary Citation of Related Structures:
2LUA - PubMed Abstract:
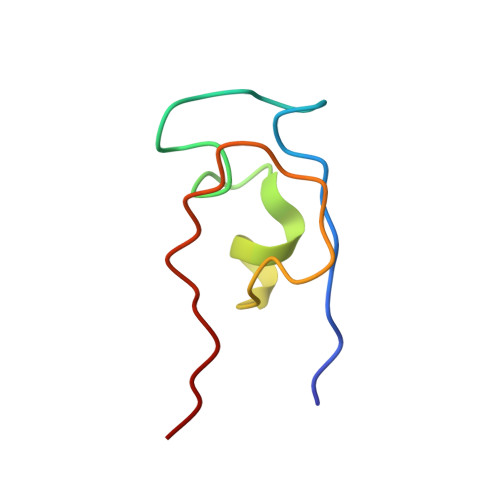
The dosage compensation complex (DCC) binds to single X chromosomes in Drosophila males and increases the transcription level of X-linked genes by approximately twofold. Male-specific lethal 2 (MSL2) together with MSL1 mediates the initial recruitment of the DCC to high-affinity sites in the X chromosome. MSL2 contains a DNA-binding cysteine-rich CXC domain that is important for X targeting. In this study, we determined the solution structure of MSL2 CXC domain by NMR spectroscopy. We identified three zinc ions in the CXC domain and determined the metal-to-cysteine connectivities from (1)H-(113)Cd correlation experiments. The structure reveals an unusual zinc-cysteine cluster composed of three zinc ions coordinated by six terminal and three bridging cysteines. The CXC domain exhibits unexpected structural homology to pre-SET motifs of histone lysine methyltransferases, expanding the distribution and structural diversity of the CXC domain superfamily. Our findings provide novel structural insight into the evolution and function of CXC domains.
- Department of Biochemistry and Molecular Biology, College of Life Sciences, Beijing Normal University, Beijing, China.
Organizational Affiliation: