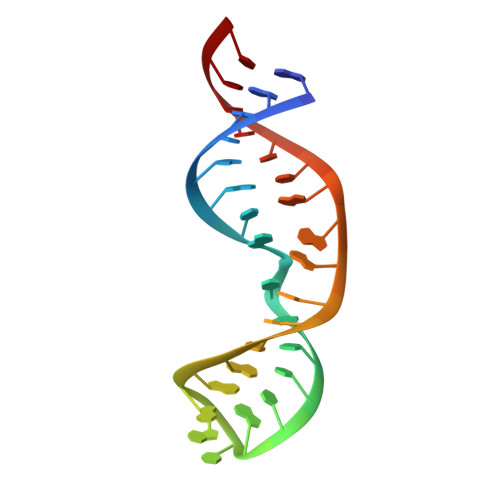
Solution structure of an alternate conformation of helix27 from Escherichia coli16S rRNA.
Spano, M.N., Walter, N.G.(2011) Biopolymers 95: 653-668
- PubMed: 21442607 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1002/bip.21626
- Primary Citation Related Structures:
2LDT - PubMed Abstract:
Helix (H)27 of 16S ribosomal (r)RNA from Escherichia coli was dubbed the "switch helix" when mutagenesis suggested that two alternative base pair registers may have distinct functional roles in the bacterial ribosome. Although more recent genetic analyses suggest that H27 conformational switching is not required for translation, previous solution studies demonstrated that the isolated E. coli H27 can dynamically convert between the 885 and 888 conformations. Here, we have solved the nuclear magnetic resonance solution structure of a locked 888 conformation. NOE and residual dipolar coupling restraints reveal an architecture that markedly differs from that of the 885 conformation found in crystal structures of the bacterial ribosome. In place of the loop E motif that characterizes the 885 conformer and that the 888 conformer cannot adopt, we find evidence for an asymmetrical A-rich internal loop stabilized by stacking interactions among the unpaired A's. Comparison of the isolated H27 888 solution structure with the 885 crystal structure within the context of the ribosome suggests a difference in overall length of H27 that presents one plausible reason for the absence of H27 conformational switching within the sterically confining ribosome.
- Department of Chemistry, University of Michigan, Ann Arbor, MI 48109-1055, USA. mnewby@clemson.edu
Organizational Affiliation: