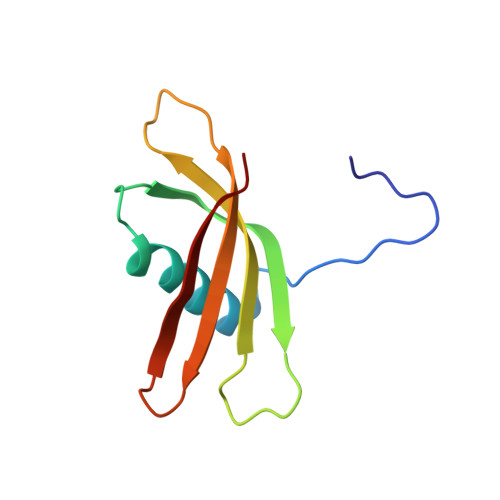
Solution structure of the squash aspartic acid proteinase inhibitor (SQAPI) and mutational analysis of pepsin inhibition.
Headey, S.J., Macaskill, U.K., Wright, M.A., Claridge, J.K., Edwards, P.J.B., Farley, P.C., Christeller, J.T., Laing, W.A., Pascal, S.M.(2010) J Biological Chem 285: 27019-27025
- PubMed: 20538608 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M110.137018
- Primary Citation Related Structures:
2KXG - PubMed Abstract:
The squash aspartic acid proteinase inhibitor (SQAPI), a proteinaceous proteinase inhibitor from squash, is an effective inhibitor of a range of aspartic proteinases. Proteinaceous aspartic proteinase inhibitors are rare in nature. The only other example in plants probably evolved from a precursor serine proteinase inhibitor. Earlier work based on sequence homology modeling suggested SQAPI evolved from an ancestral cystatin. In this work, we determined the solution structure of SQAPI using NMR and show that SQAPI shares the same fold as a plant cystatin. The structure is characterized by a four-strand anti-parallel beta-sheet gripping an alpha-helix in an analogous manner to fingers of a hand gripping a tennis racquet. Truncation and site-specific mutagenesis revealed that the unstructured N terminus and the loop connecting beta-strands 1 and 2 are important for pepsin inhibition, but the loop connecting strands 3 and 4 is not. Using ambiguous restraints based on the mutagenesis results, SQAPI was then docked computationally to pepsin. The resulting model places the N-terminal strand of SQAPI in the S' side of the substrate binding cleft, whereas the first SQAPI loop binds on the S side of the cleft. The backbone of SQAPI does not interact with the pepsin catalytic Asp(32)-Asp(215) diad, thus avoiding cleavage. The data show that SQAPI does share homologous structural elements with cystatin and appears to retain a similar protease inhibitory mechanism despite its different target. This strongly supports our hypothesis that SQAPI evolved from an ancestral cystatin.
- Institute of Fundamental Sciences, Massey University, Palmerston North 4442, New Zealand.
Organizational Affiliation: