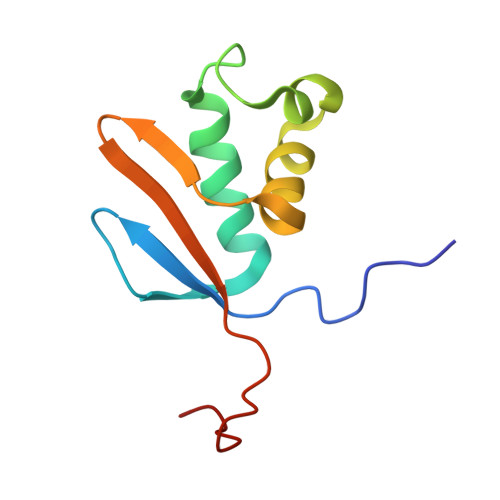
Solution NMR structure of Asl3597 from Nostoc sp. PCC7120, the first structure from protein domain family PF12095, reveals a novel fold.
Feldmann, E.A., Ramelot, T.A., Yang, Y., Xiao, R., Acton, T.B., Everett, J.K., Montelione, G.T., Kennedy, M.A.(2012) Proteins 80: 671-675
- PubMed: 22113821
- DOI: https://doi.org/10.1002/prot.23236
- Primary Citation of Related Structures:
2KRX - PubMed Abstract:
The protein domain family PF12095 (DUF3571) is a functionally uncharacterized family of small proteins conserved from cyanobacteria to plants that are typically 85 to 95 amino acids in length in cyanobacteria. In this report, we describe the solution NMR structure of the 86-residue protein Asl3597 from Nostoc sp. PCC7120. The structure of Asl3597, which constitutes the first three-dimensional structure from protein family PF12095, has a unique α/β sandwich fold consisting of four anti-parallel β-strands opposite three continuous α-helices. Asl3597 may have a role in the assembly of the hydrophilic subcomplex of the cyanobacterial NAD(P)H complex as suggested by data for the orthologous Chlororespiratory reduction 7 protein from Arabidopsis thaliana.
- Department of Chemistry and Biochemistry and the Northeast Structural Genomics Consortium, Miami University, Oxford, Ohio 45056.
Organizational Affiliation: