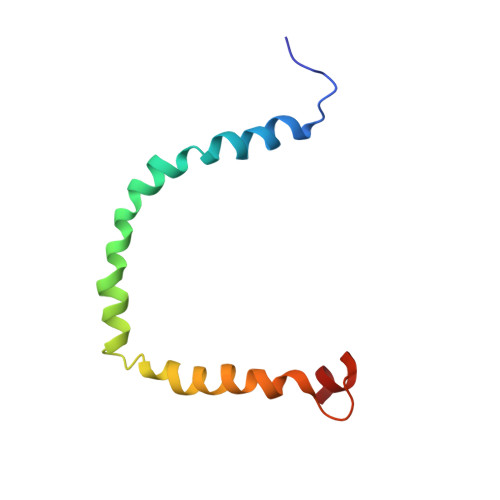
Structure and Dynamics of Human Apolipoprotein CIII
Gangabadage, C.S., Zdunek, J., Tessari, M., Nilsson, S., Olivecrona, G., Wijmenga, S.S.(2008) J Biological Chem 283: 17416-17427
- PubMed: 18408013 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M800756200
- Primary Citation Related Structures:
2JQ3 - PubMed Abstract:
Human apolipoprotein CIII (apoCIII) is a surface component of chylomicrons, very low density lipoproteins, and high density lipoproteins. ApoCIII inhibits lipoprotein lipase as well as binding of lipoproteins to cell surface heparan sulfate proteoglycans and receptors. High levels of apoCIII are often correlated with elevated levels of blood lipids (hypertriglyceridemia). Here, we report the three-dimensional NMR structure and dynamics of human apo-CIII in complex with SDS micelles, mimicking its natural lipid-bound state. Thanks to residual dipolar coupling data, the first detailed view is obtained of the structure and dynamics of an intact apolipoprotein in its lipid-bound state. ApoCIII wraps around the micelle surface as a necklace of six approximately 10-residue amphipathic helices, which are curved and connected via semiflexible hinges. Three positively charged (Lys) residues line the polar faces of helices 1 and 2. Interestingly, their three-dimensional conformation is similar to that of the low density lipoprotein receptor binding motifs of apoE/B and the receptor-associated protein. At the C-terminal side of apoCIII, an array of negatively charged residues lines the polar faces of helices 4 and 5 and the adjacent flexible loop. Sequence comparison shows that this asymmetric charge distribution along the solvent-exposed face of apoCIII as well as other structural features are conserved among mammals. This structure provides a template for exploration of molecular mechanisms by which human apoCIII inhibits lipoprotein lipase and receptor binding.
- Department of Physical Chemistry-Biophysical Chemistry, Radboud University of Nijmegen, Toernooiveld 1, 6525 ED, Nijmegen, The Netherlands.
Organizational Affiliation: