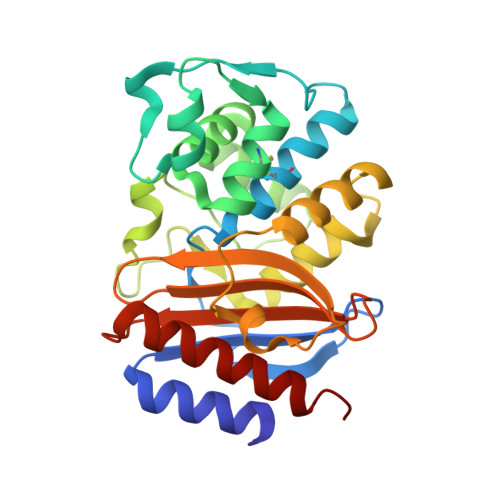
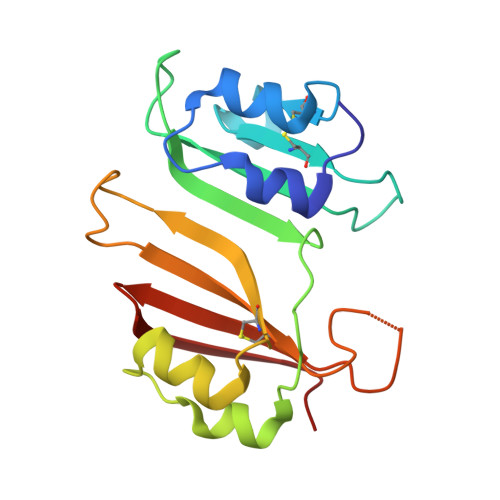
Structural and Computational Characterization of the SHV-1 beta-Lactamase-beta-Lactamase Inhibitor Protein Interface.
Reynolds, K.A., Thomson, J.M., Corbett, K.D., Bethel, C.R., Berger, J.M., Kirsch, J.F., Bonomo, R.A., Handel, T.M.(2006) J Biological Chem 281: 26745-26753
- PubMed: 16809340 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M603878200
- Primary Citation Related Structures:
2G2U, 2G2W - PubMed Abstract:
Beta-lactamase inhibitor protein (BLIP) binds a variety of class A beta-lactamases with affinities ranging from micromolar to picomolar. Whereas the TEM-1 and SHV-1 beta-lactamases are almost structurally identical, BLIP binds TEM-1 approximately 1000-fold tighter than SHV-1. Determining the underlying source of this affinity difference is important for understanding the molecular basis of beta-lactamase inhibition and mechanisms of protein-protein interface specificity and affinity. Here we present the 1.6A resolution crystal structure of SHV-1.BLIP. In addition, a point mutation was identified, SHV D104E, that increases SHV.BLIP binding affinity from micromolar to nanomolar. Comparison of the SHV-1.BLIP structure with the published TEM-1.BLIP structure suggests that the increased volume of Glu-104 stabilizes a key binding loop in the interface. Solution of the 1.8A SHV D104K.BLIP crystal structure identifies a novel conformation in which this binding loop is removed from the interface. Using these structural data, we evaluated the ability of EGAD, a program developed for computational protein design, to calculate changes in the stability of mutant beta-lactamase.BLIP complexes. Changes in binding affinity were calculated within an error of 1.6 kcal/mol of the experimental values for 112 mutations at the TEM-1.BLIP interface and within an error of 2.2 kcal/mol for 24 mutations at the SHV-1.BLIP interface. The reasonable success of EGAD in predicting changes in interface stability is a promising step toward understanding the stability of the beta-lactamase.BLIP complexes and computationally assisted design of tight binding BLIP variants.
- Skaggs School of Pharmacy and Pharmaceutical Sciences, University of California San Diego, La Jolla, California 92093-0684, USA.
Organizational Affiliation: