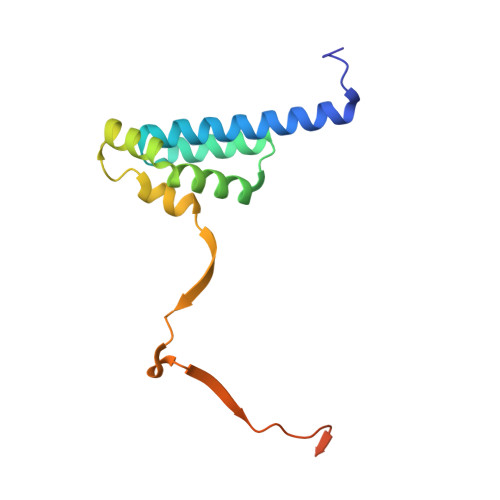
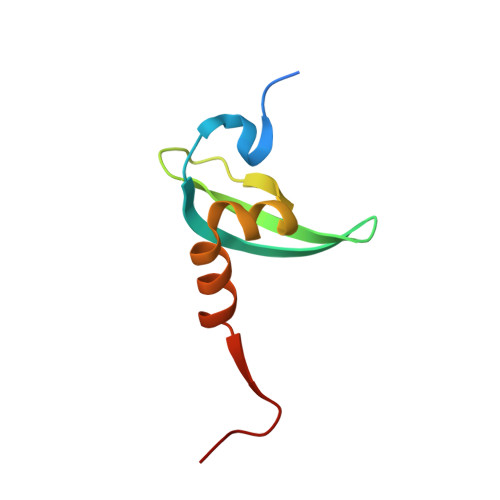
Crystal structure of the HP1-EMSY complex reveals an unusual mode of HP1 binding.
Huang, Y., Myers, M.P., Xu, R.M.(2006) Structure 14: 703-712
- PubMed: 16615912 Search on PubMed
- DOI: https://doi.org/10.1016/j.str.2006.01.007
- Primary Citation Related Structures:
2FMM - PubMed Abstract:
Heterochromatin protein-1 (HP1) plays an essential role in both the assembly of higher-order chromatin structure and epigenetic inheritance. The C-terminal chromo shadow domain (CSD) of HP1 is responsible for homodimerization and interaction with a number of chromatin-associated nonhistone proteins, including EMSY, which is a BRCA2-interacting protein that has been implicated in the development of breast and ovarian cancer. We have determined the crystal structure of the HP1beta CSD in complex with the N-terminal domain of EMSY at 1.8 A resolution. Surprisingly, the structure reveals that EMSY is bound by two HP1 CSD homodimers, and the binding sequences differ from the consensus HP1 binding motif PXVXL. This structural information expands our understanding of HP1 binding specificity and provides insights into interactions between HP1 homodimers that are likely to be important for heterochromatin formation.
- Cold Spring Harbor Laboratory, Cold Spring Harbor, New York 11724, USA.
Organizational Affiliation: