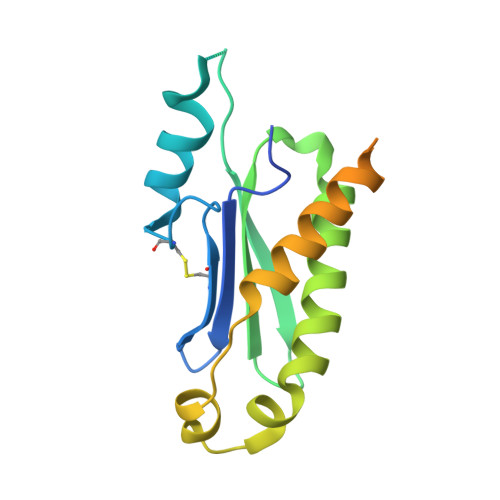
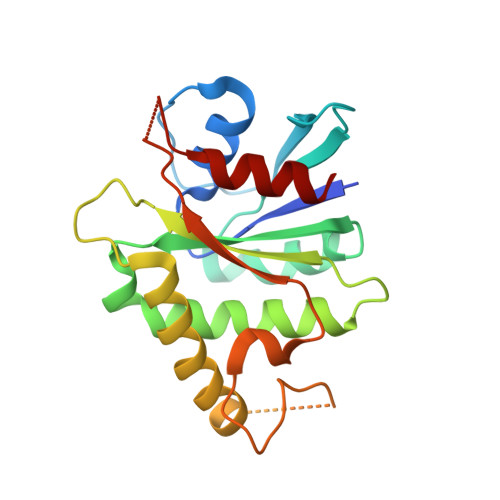
The structure of the mammalian signal recognition particle (SRP) receptor as prototype for the interaction of small GTPases with Longin domains.
Schlenker, O., Hendricks, A., Sinning, I., Wild, K.(2006) J Biological Chem 281: 8898-8906
- PubMed: 16439358 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M512415200
- Primary Citation Related Structures:
2FH5 - PubMed Abstract:
The eukaryotic signal recognition particle (SRP) and its receptor (SR) play a central role in co-translational targeting of secretory and membrane proteins to the endoplasmic reticulum. The SR is a heterodimeric complex assembled by the two GTPases SRalpha and SRbeta, which is membrane-anchored. Here we present the 2.45-A structure of mammalian SRbeta in its Mg2+ GTP-bound state in complex with the minimal binding domain of SRalpha termed SRX. SRbeta is a member of the Ras-GTPase superfamily closely related to Arf and Sar1, while SRX belongs to the SNARE-like superfamily with a fold also known as longin domain. SRX binds to the P loop and the switch regions of SRbeta-GTP. The binding mode and structural similarity with other GTPase-effector complexes suggests a co-GAP (GTPase-activating protein) function for SRX. Comparison with the homologous yeast structure and other longin domains reveals a conserved adjustable hydrophobic surface within SRX which is of central importance for the SRbeta-GTP:SRX interface. A helix swap in SRX results in the formation of a dimer in the crystal structure. Based on structural conservation we present the SRbeta-GTP:SRX structure as a prototype for conserved interactions in a variety of GTPase regulated targeting events occurring at endomembranes.
- Biochemie-Zentrum der Universität Heidelberg (BZH), Im Neuenheimer Feld 328, D-69120 Heidelberg, Germany.
Organizational Affiliation: