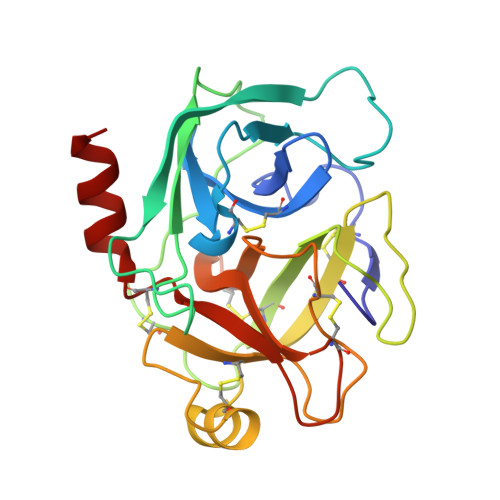
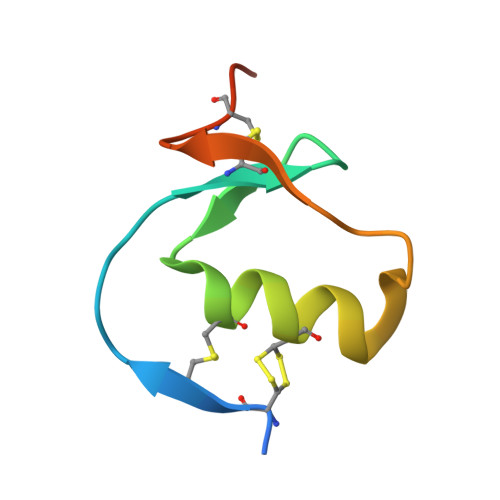
The Kazal-type inhibitors infestins 1 and 4 differ in specificity but are similar in three-dimensional structure.
Campos, I.T., Souza, T.A., Torquato, R.J., De Marco, R., Tanaka-Azevedo, A.M., Tanaka, A.S., Barbosa, J.A.R.G.(2012) Acta Crystallogr D Biol Crystallogr 68: 695-702
- PubMed: 22683792 Search on PubMed
- DOI: https://doi.org/10.1107/S0907444912009067
- Primary Citation Related Structures:
2ERW, 2F3C - PubMed Abstract:
Blood coagulation is an important process in haemostasis, and disorders of blood coagulation can lead to an increased risk of haemorrhage and thrombosis. Coagulation is highly conserved in mammals and has been comprehensively studied in humans in the investigation of bleeding or thrombotic diseases. Some substances can act as inhibitors of blood coagulation and may affect one or multiple enzymes throughout the process. A specific thrombin inhibitor called infestin has been isolated from the midgut of the haematophagous insect Triatoma infestans. Infestin is a member of the nonclassical Kazal-type serine protease inhibitors and is composed of four domains, all of which have a short central α-helix and a small antiparallel β-sheet. Domains 1 and 4 of infestin (infestins 1 and 4) possess specific inhibitory activities. Infestin 1 inhibits thrombin, while infestin 4 is an inhibitor of factor XIIa, plasmin and factor Xa. Here, the structure determination and structural analysis of infestin 1 complexed with trypsin and of infestin 4 alone are reported. Through molecular modelling and docking, it is suggested that the protein-protein binding site is conserved in the infestin 1-thrombin complex compared with other Kazal-type inhibitors. Infestin 4 is able to bind factor XIIa, and the F9N and N11R mutants selected by phage display were shown to be more selective for factor XIIa in comparison to the wild type.
- Departamento de Bioquímica, Escola Paulista de Medicina, Universidade Federal de São Paulo, Rua Três de Maio 100, 04044-020 São Paulo-SP, Brazil.
Organizational Affiliation: