photo-activation state of Fe-type NHase with n-BA in anaerobic condition
Kawano, Y., Hashimoto, K., Odaka, M., Nakayama, H., Takio, K., Endo, I., Kamiya, N.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Nitrile hydratase subunit alpha | 206 | Rhodococcus erythropolis | Mutation(s): 2 EC: 4.2.1.84 | 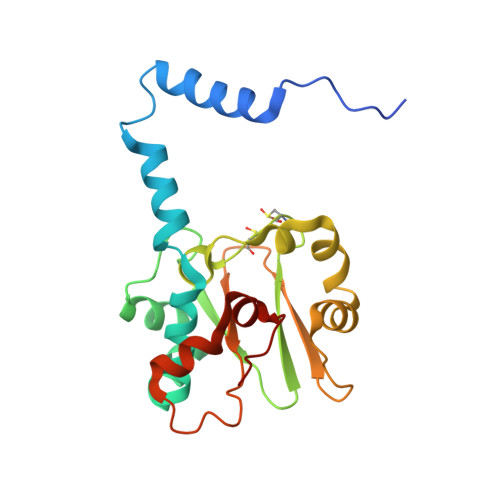 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P13448 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Nitrile hydratase subunit beta | 212 | Rhodococcus erythropolis | Mutation(s): 0 EC: 4.2.1.84 | 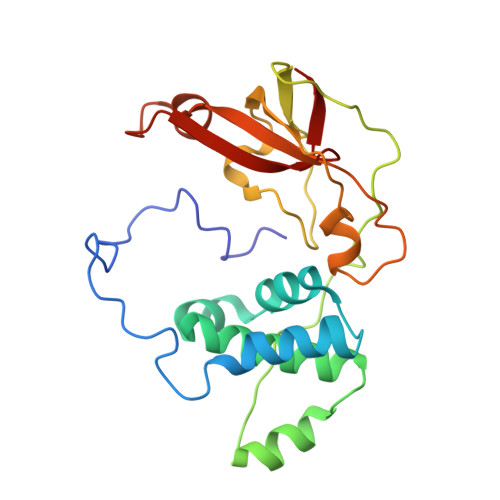 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P13449 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| BUA Download:Ideal Coordinates CCD File | D [auth A] | butanoic acid C4 H8 O2 FERIUCNNQQJTOY-UHFFFAOYSA-N |  | ||
| FE Download:Ideal Coordinates CCD File | C [auth A] | FE (III) ION Fe VTLYFUHAOXGGBS-UHFFFAOYSA-N |  | ||
| MG Download:Ideal Coordinates CCD File | E [auth B] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Modified Residues 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| CSD Query on CSD | A | L-PEPTIDE LINKING | C3 H7 N O4 S |  | CYS |
| CSO Query on CSO | A | L-PEPTIDE LINKING | C3 H7 N O3 S |  | CYS |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 115.329 | α = 90 |
| b = 60.792 | β = 124.96 |
| c = 82.024 | γ = 90 |
| Software Name | Purpose |
|---|---|
| SHELX | model building |
| SHELXL-97 | refinement |
| HKL-2000 | data reduction |
| SCALEPACK | data scaling |
| CNS | phasing |