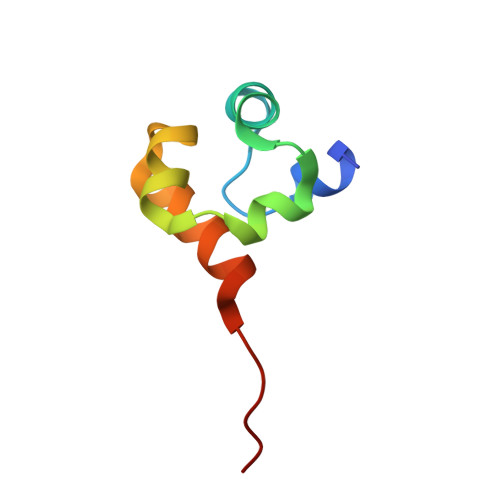
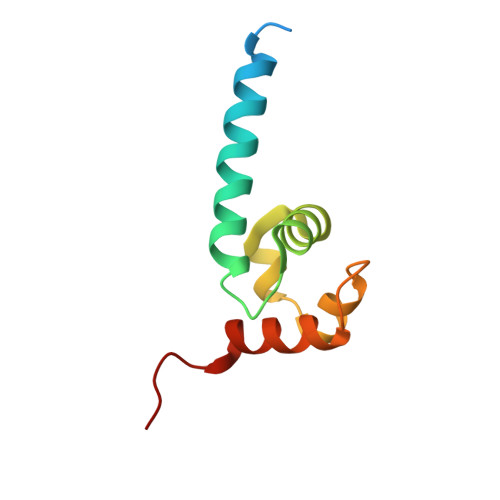
Crystal structure and DNA binding functions of ERCC1, a subunit of the DNA structure-specific endonuclease XPF-ERCC1.
Tsodikov, O.V., Enzlin, J.H., Scharer, O.D., Ellenberger, T.(2005) Proc Natl Acad Sci U S A 102: 11236-11241
- PubMed: 16076955
- DOI: https://doi.org/10.1073/pnas.0504341102
- Primary Citation of Related Structures:
2A1I, 2A1J - PubMed Abstract:
Human XPF-ERCC1 is a DNA endonuclease that incises a damaged DNA strand on the 5' side of a lesion during nucleotide excision repair and has additional role(s) in homologous recombination and DNA interstrand crosslink repair. We show that a truncated form of XPF lacking the N-terminal helicase-like domain in complex with ERCC1 exhibits a structure-specific endonuclease activity with similar specificity to that of full-length XPF-ERCC1. Two domains of ERCC1, a central domain and a C-terminal tandem helix-hairpin-helix (HhH2) dimerization domain, bind to ssDNA. The central domain of ERCC1 binds ssDNA/dsDNA junctions with a defined polarity, preferring a 5' single-stranded overhang. The XPF-ERCC1 HhH2 domain heterodimer contains two independent ssDNA-binding surfaces, which are revealed by a crystal structure of the protein complex. A crystal structure of the central domain of ERCC1 shows its fold is strikingly similar to that of the nuclease domains of the archaeal Mus81/XPF homologs, despite very low sequence homology. A groove lined with basic and aromatic residues on the surface of ERCC1 has apparently been adapted to interact with ssDNA. On the basis of these crystallographic and biochemical studies, we propose a model in which XPF-ERCC1 recognizes a branched DNA substrate by binding the two ssDNA arms with the two HhH2 domains of XPF and ERCC1 and by binding the 5'-ssDNA arm with the central domain of ERCC1.
- Department of Biological Chemistry and Molecular Pharmacology, Harvard Medical School, 240 Longwood Avenue, Boston, MA 02115, USA.
Organizational Affiliation: