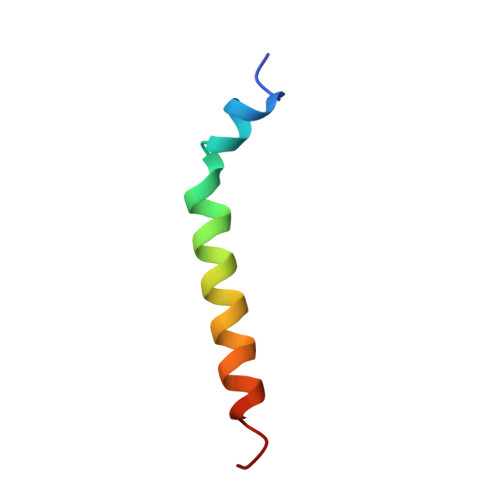
Solution structure of a recombinant type I sculpin antifreeze protein
Kwan, A.H.Y., Fairley, K., Anderberg, P.I., Liew, C.W., Harding, M.M., Mackay, J.P.(2005) Biochemistry 44: 1980-1988
- PubMed: 15697223 Search on PubMed
- DOI: https://doi.org/10.1021/bi047782j
- Primary Citation Related Structures:
1Y03, 1Y04 - PubMed Abstract:
We have determined the solution structure of rSS3, a recombinant form of the type I shorthorn sculpin antifreeze protein (AFP), at 278 and 268 K. This AFP contains an unusual sequence of N-terminal residues, together with two of the 11-residue repeats that are characteristic of the type I winter flounder AFP. The solution conformation of the N-terminal region of the sculpin AFP has been assumed to be the critical factor that results in recognition of different ice planes by the sculpin and flounder AFPs. At 278 K, the two repeats units (residues 11-20 and 21-32) in rSS3 form a continuous alpha-helix, with the residues 30-33 in the second repeat somewhat less well defined. Within the N-terminal region, residues 2-6 are well defined and helical and linked to the main helix by a more flexible region comprising residues A7-T11. At 268 K the AFP is overall more helical but retains the apparent hinge region. The helical conformation of the two repeats units is almost identical to the corresponding repeats in the type I winter flounder AFP. We also show that while tetracetylated rSS3 has antifreeze activity comparable to the natural AFP, its overall structure is the same as that of the unacetylated peptide. These data provide some insight into the structural determinants of antifreeze activity and should assist in the development of models that explain the recognition of different ice interfaces by the sculpin and flounder type I AFPs.
- School of Chemistry, The University of Sydney, NSW 2006, Australia.
Organizational Affiliation: