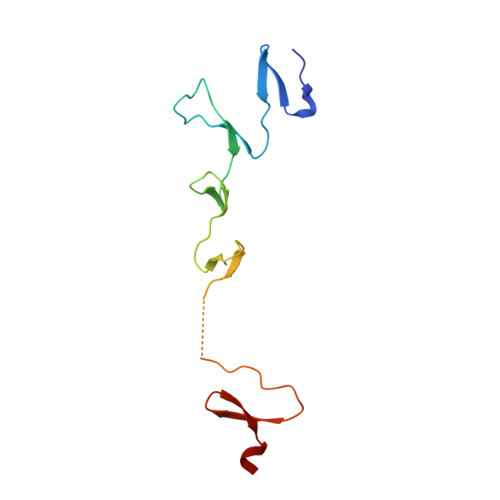
Adenovirus Fibre Shaft Sequences Fold Into the Native Triple Beta-Spiral Fold When N-Terminally Fused to the Bacteriophage T4 Fibritin Foldon Trimerisation Motif
Papanikolopoulou, K., Teixeira, S., Belrhali, H., Forsyth, V.T., Mitraki, A., van Raaij, M.J.(2004) J Mol Biology 342: 219
- PubMed: 15313619 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2004.07.008
- Primary Citation Related Structures:
1V1H, 1V1I - PubMed Abstract:
Adenovirus fibres are trimeric proteins that consist of a globular C-terminal domain, a central fibrous shaft and an N-terminal part that attaches to the viral capsid. In the presence of the globular C-terminal domain, which is necessary for correct trimerisation, the shaft segment adopts a triple beta-spiral conformation. We have replaced the head of the fibre by the trimerisation domain of the bacteriophage T4 fibritin, the foldon. Two different fusion constructs were made and crystallised, one with an eight amino acid residue linker and one with a linker of only two residues. X-ray crystallographic studies of both fusion proteins shows that residues 319-391 of the adenovirus type 2 fibre shaft fold into a triple beta-spiral fold indistinguishable from the native structure, although this is now resolved at a higher resolution of 1.9 A. The foldon residues 458-483 also adopt their natural structure. The intervening linkers are not well ordered in the crystal structures. This work shows that the shaft sequences retain their capacity to fold into their native beta-spiral fibrous fold when fused to a foreign C-terminal trimerisation motif. It provides a structural basis to artificially trimerise longer adenovirus shaft segments and segments from other trimeric beta-structured fibre proteins. Such artificial fibrous constructs, amenable to crystallisation and solution studies, can offer tractable model systems for the study of beta-fibrous structure. They can also prove useful for gene therapy and fibre engineering applications.
- Institut de Biologie Structurale Jean-Pierre Ebel, 41 rue Jules Horowitz, F-38042 Grenoble, France.
Organizational Affiliation: