Crystal structure of Mycobacterium tuberculosis citrate lyase beta subunit and its unusual triphosphate binding site
Goulding, C.W., Lerkin, T., Kim, C.Y., Segelke, B., Terwilliger, T., Eisenberg, E.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| citE | 273 | Mycobacterium tuberculosis | Mutation(s): 6 Gene Names: Rv2498c EC: 4.1.3.6 (PDB Primary Data), 4.1 (UniProt) | 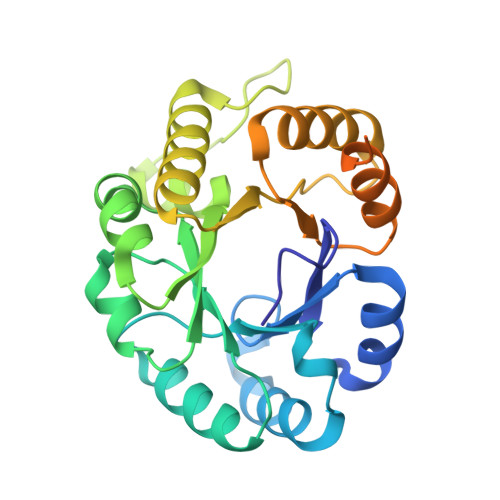 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P9WPE1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ATP Download:Ideal Coordinates CCD File | B [auth A] | ADENOSINE-5'-TRIPHOSPHATE C10 H16 N5 O13 P3 ZKHQWZAMYRWXGA-KQYNXXCUSA-N |  | ||
| FMT Download:Ideal Coordinates CCD File | C [auth A] | FORMIC ACID C H2 O2 BDAGIHXWWSANSR-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 91.214 | α = 90 |
| b = 91.214 | β = 90 |
| c = 220.233 | γ = 120 |
| Software Name | Purpose |
|---|---|
| CNS | refinement |
| DENZO | data reduction |
| SCALEPACK | data scaling |
| EPMR | phasing |